Advanced Physics → Advanced Thermal Physics → Thermodynamic Processes
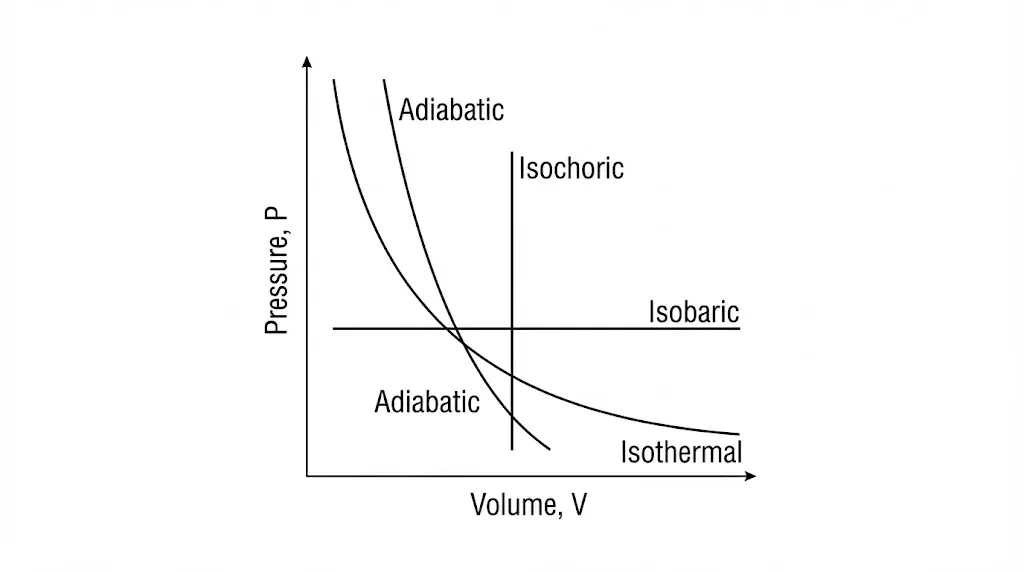
A thermodynamic process defines a specific path between equilibrium states, governed by heat transfer, work done, and internal energy change.
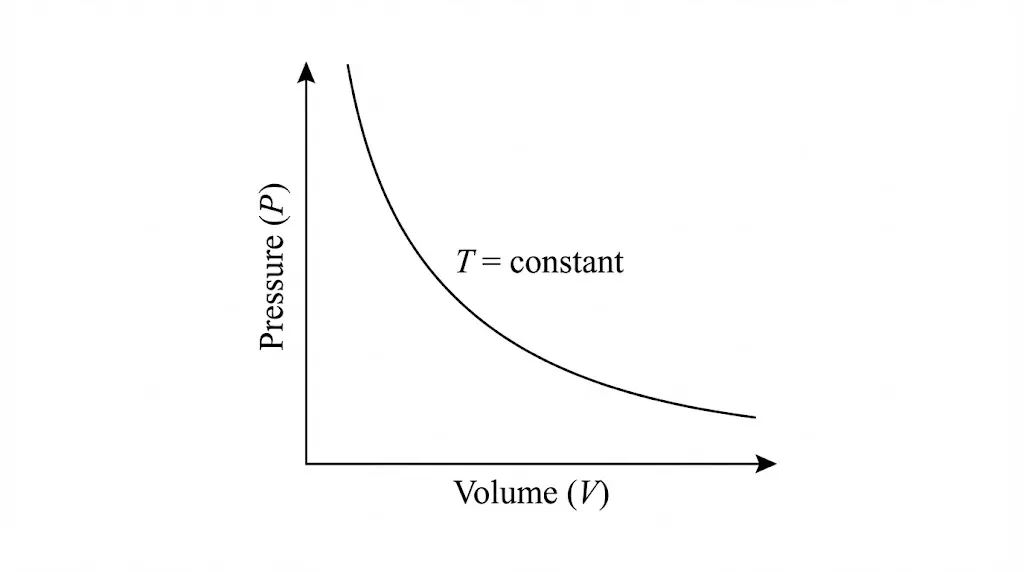
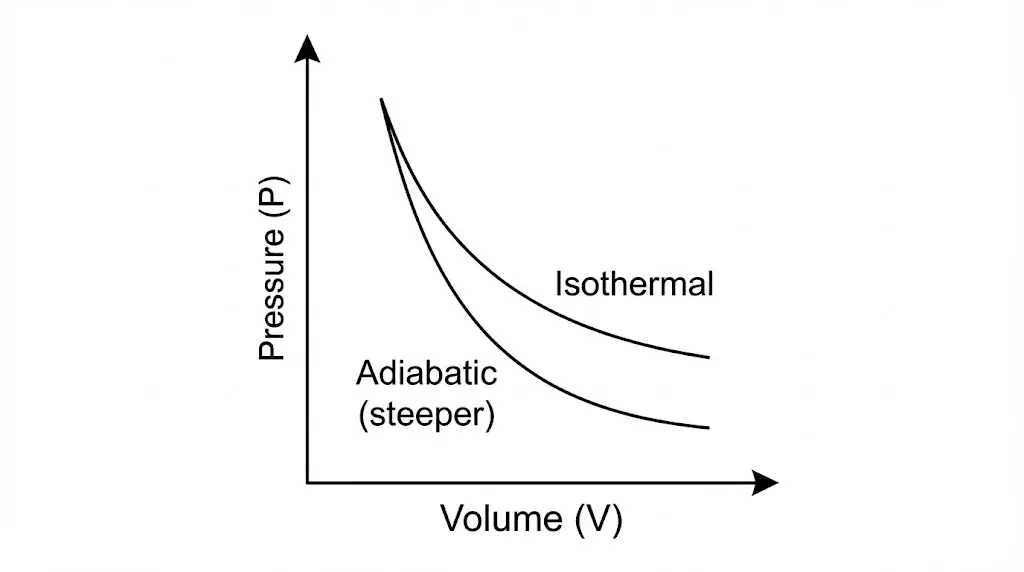
1. Isothermal Process
In an isothermal process, temperature remains constant. Heat exchange with the surroundings compensates exactly for work done by or on the system.
![]()
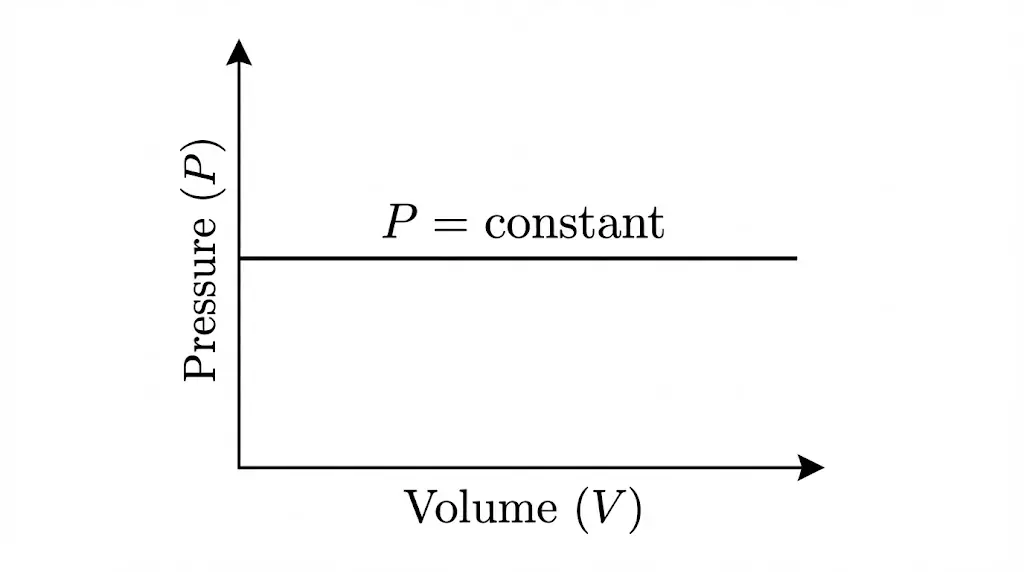
2. Isobaric Process
In an isobaric process, pressure remains constant. The system expands or contracts as temperature changes.
![]()
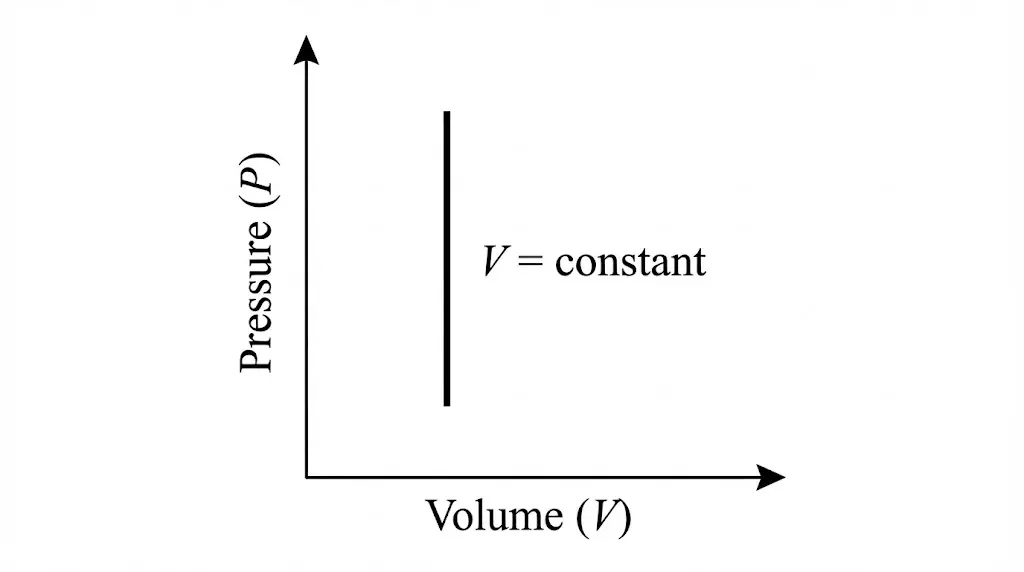
3. Isochoric Process
In an isochoric process, volume remains constant. No work is done, and all heat supplied changes internal energy.
![]()
4. Adiabatic Process
An adiabatic process occurs without heat exchange. Changes in internal energy arise solely from work done.
![]()
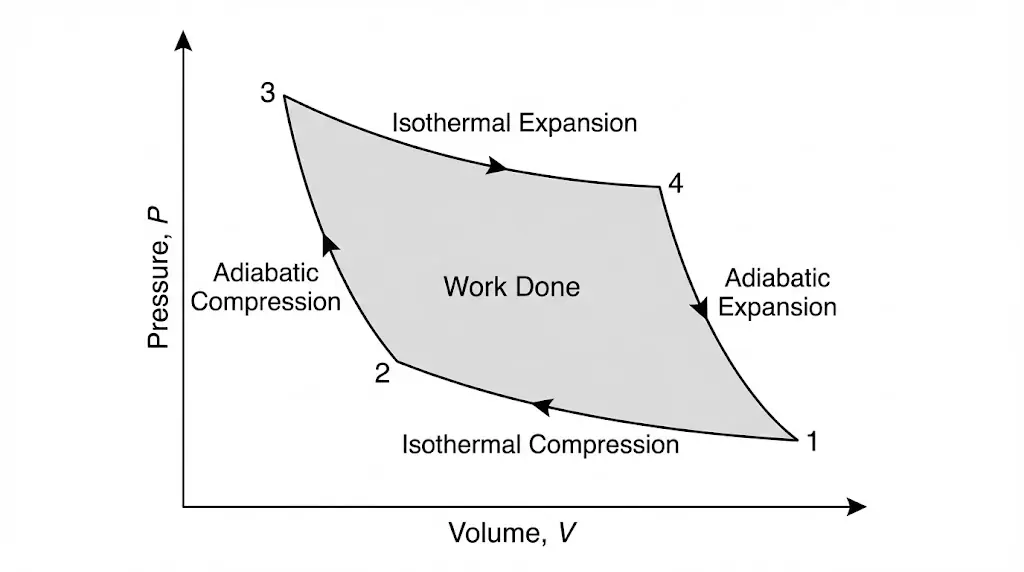
5. Cyclic Process
A cyclic process returns the system to its initial state. Net work done equals the net heat exchanged over the cycle.
Level 1 — Conceptual
Problem 1. Define constraint motion. Give one everyday example.
Solution
Constraint motion refers to the motion of objects that are connected or restricted in such a way that the motion of one object affects the motion of another.
Example: Two blocks connected by a string over a pulley.
Problem 2. Why do two blocks connected by an inextensible string have the same magnitude of acceleration?
Solution
An inextensible string does not stretch, so any displacement of one block must produce an equal displacement of the other block in the opposite direction. Hence, their accelerations are equal in magnitude.
Level 2 – Numerical
Problem 3. Two blocks of masses 2 kg and 3 kg are connected by a light string over a smooth pulley. Find the acceleration of the system. (Take g = 10 m/s²)
Solution
Net driving force = (3 − 2)g = 1 × 10 = 10 N
Total mass = 2 + 3 = 5 kg
Acceleration,
a = F / m = 10 / 5 = 2 m/s²
Problem 4. A block rests on a smooth table and is connected to a hanging mass. Explain why both masses have the same acceleration.
Solution
The string is light and inextensible, so its length remains constant. Any motion of one mass immediately produces motion in the other, resulting in the same acceleration for both masses.
Level 3 — Advanced
Problem 5. In a system of three blocks connected in a straight line on a smooth surface, an external force F is applied to the last block. Find the acceleration of the system.
Solution
Let the masses be m₁, m₂, and m₃.
Total mass of the system = m₁ + m₂ + m₃
Using Newton’s second law for the whole system:
a = F / (m₁ + m₂ + m₃)
