Advanced Physics → Advanced Thermal Physics → Entropy and Laws of Thermodynamics
Entropy explains the direction of natural processes and why some processes are irreversible.
1. Why Entropy Was Needed
The first law of thermodynamics accounts for energy conservation but does not explain why certain processes occur spontaneously.
Entropy introduces a direction to physical processes.
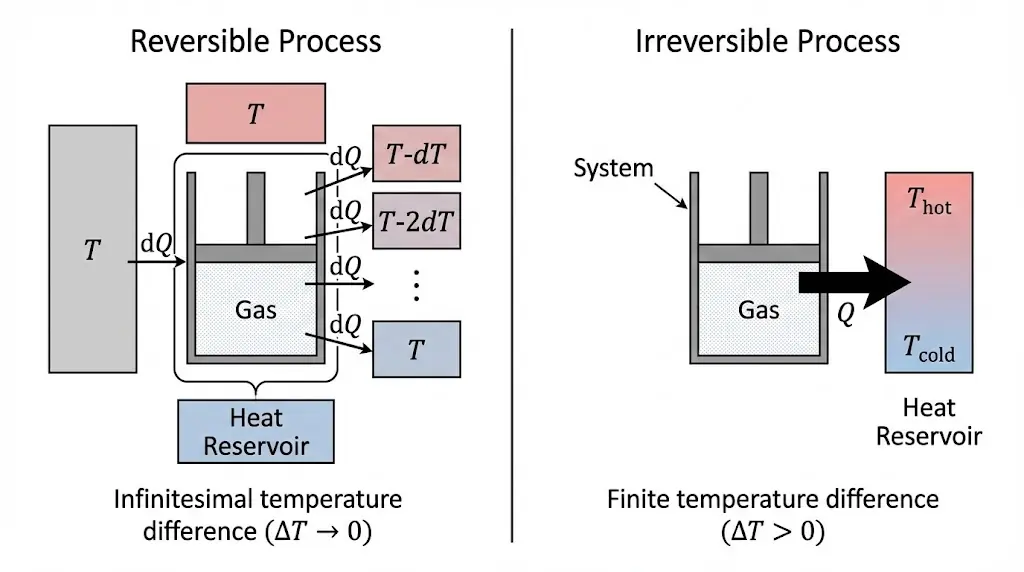
2. Reversible and Irreversible Processes
A reversible process occurs infinitely slowly and can be reversed without leaving changes in the surroundings.
3. Definition of Entropy
For a reversible process, entropy change is defined as:
![]()
This matches the thermodynamic definition of entropy as heat absorbed reversibly divided by temperature.
Entropy is a state function, meaning its value depends only on the thermodynamic state of the system and not on the path taken. Although the definition uses a reversible process, entropy itself is defined for all real processes.
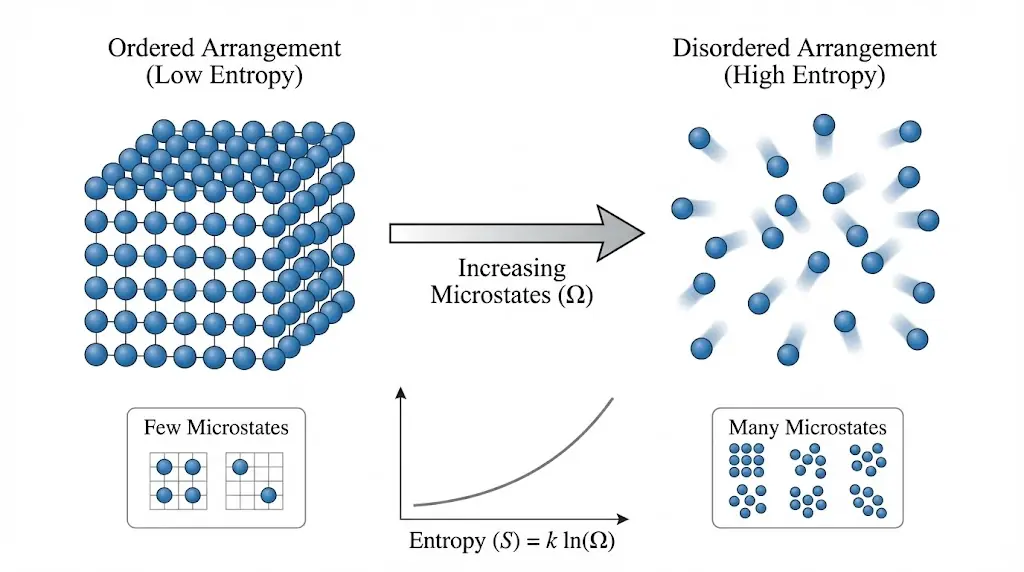
Physically, entropy measures the extent to which energy is spread out or dispersed among the available microscopic configurations of a system.
A higher entropy state corresponds to a larger number of possible microscopic arrangements (microstates) that result in the same macroscopic appearance.
4. Entropy Change in Isothermal Expansion
For a reversible isothermal expansion of an ideal gas:
![]()
This result follows from integrating ![]() with
with ![]() .
.
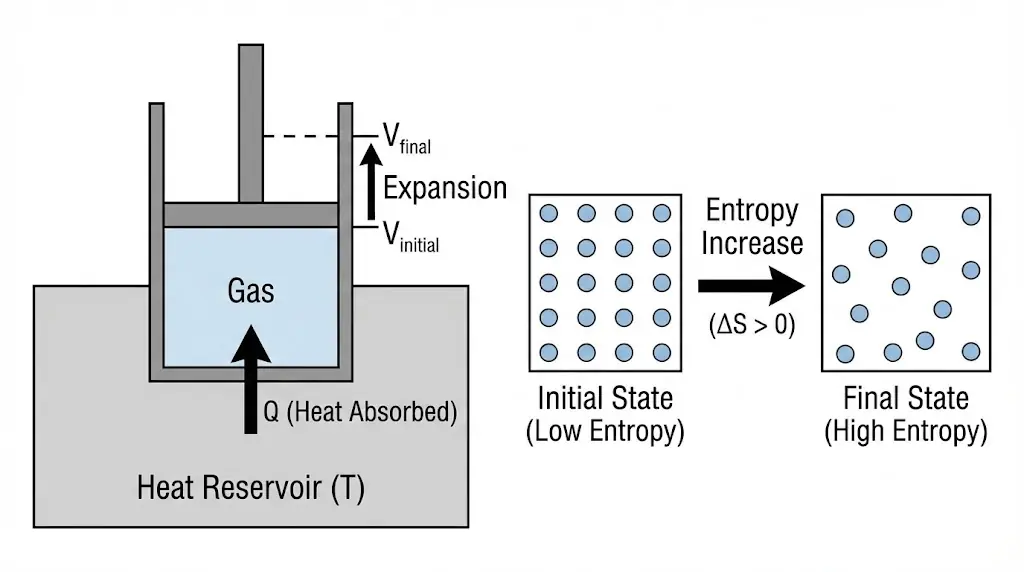
During isothermal expansion, the temperature remains constant, but the volume available to gas molecules increases.
This increase in volume allows molecules to occupy a larger number of spatial configurations, increasing the number of accessible microstates.
Hence, even though internal energy remains unchanged, entropy increases due to increased molecular disorder.
5. Second Law of Thermodynamics
The entropy of an isolated system never decreases.
![]()
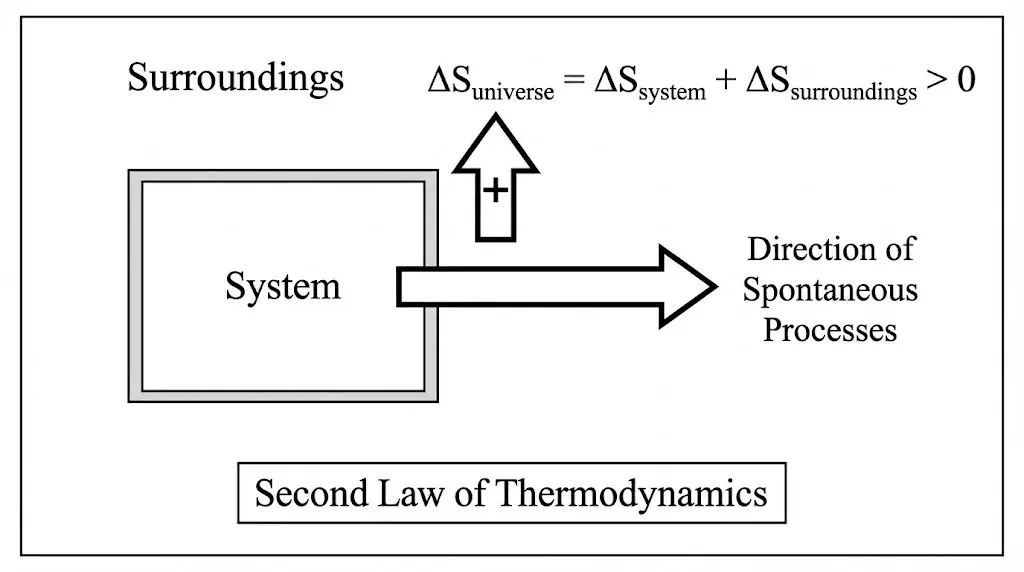
The second law introduces irreversibility into thermodynamics. While energy conservation allows many processes in principle, the entropy principle selects which processes can actually occur.
Any spontaneous process increases the entropy of the universe, even if the entropy of a particular system decreases.
This law explains why heat flows naturally from hot to cold bodies and never in the reverse direction without external intervention.
6. Entropy and Heat Engines
Heat engines operate by transferring heat between reservoirs. Entropy analysis reveals efficiency limits.
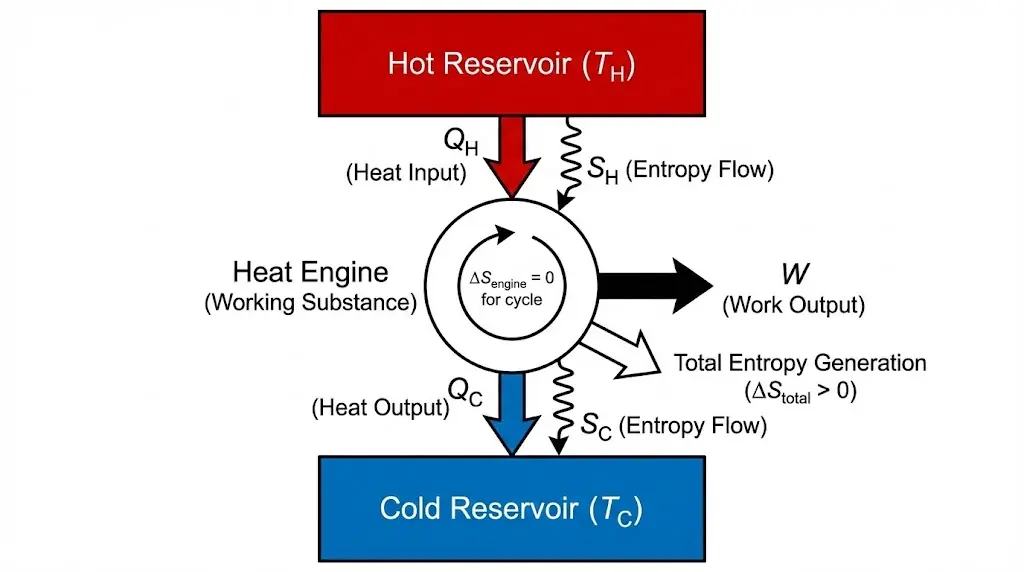
In heat engines, entropy analysis reveals that not all heat absorbed from the hot reservoir can be converted into useful work.
Some entropy must always be rejected to the cold reservoir, which places an upper limit on engine efficiency.
This entropy requirement explains why perpetual motion machines of the second kind are impossible.
7. Entropy and the Arrow of Time
Macroscopic irreversibility arises from microscopic probability. Entropy provides the physical meaning of time’s arrow.
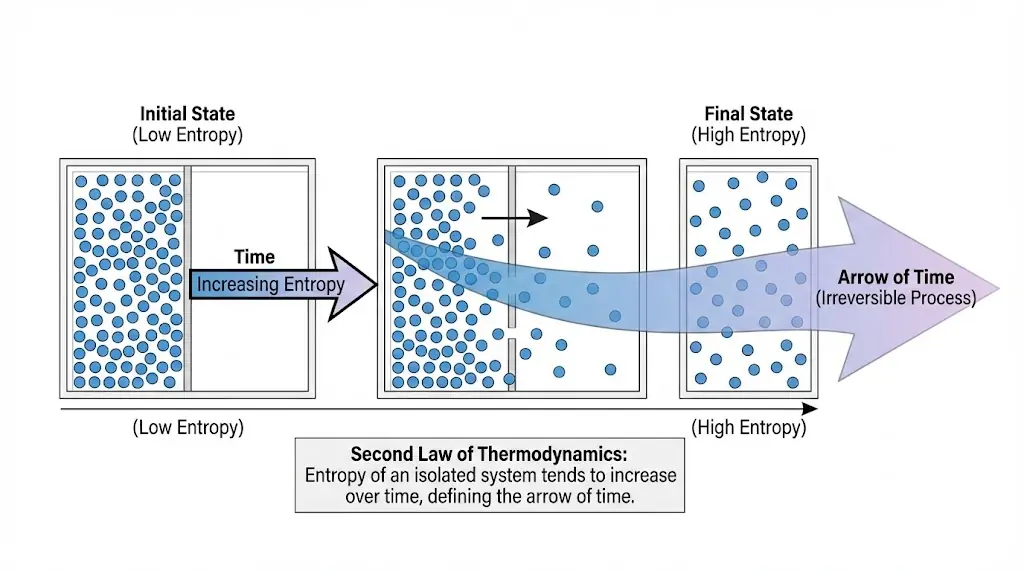
Microscopic physical laws are largely time-reversible, yet macroscopic processes show a clear direction in time.
Entropy provides the bridge between these two descriptions. Processes evolve from less probable states to more probable ones, giving rise to an apparent arrow of time.
This statistical interpretation explains why broken objects do not spontaneously reassemble and why mixing processes do not reverse naturally.
