Advanced Physics → Advanced Thermal Physics → Heat Engines & Refrigerators
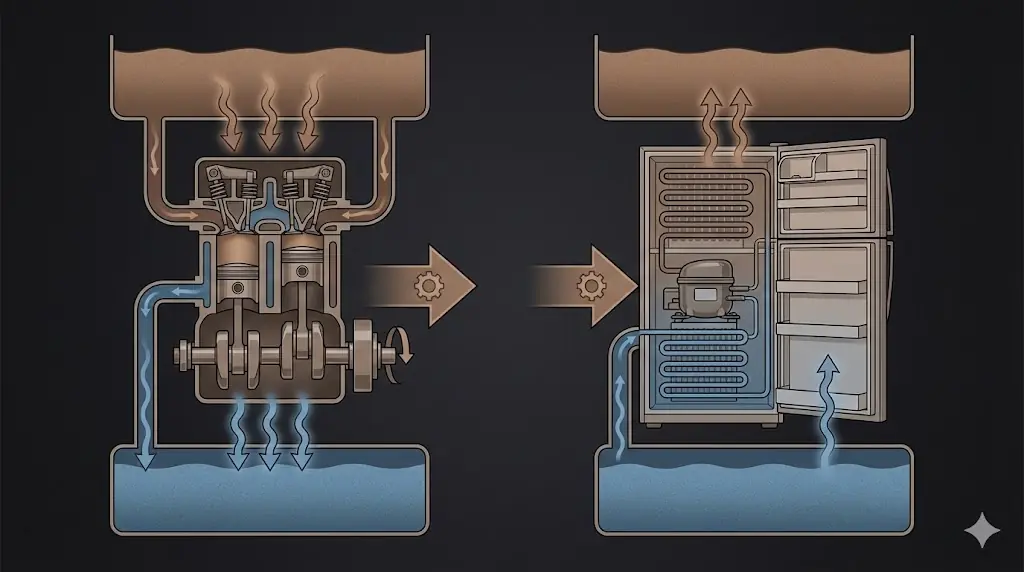
Central idea
The second law of thermodynamics limits how efficiently heat can be converted into work.
The second law of thermodynamics limits how efficiently heat can be converted into work.
1. What Is a Heat Engine?
A heat engine is a cyclic device that absorbs heat from a hot reservoir, converts part of it into work, and rejects the remaining heat to a cold reservoir.
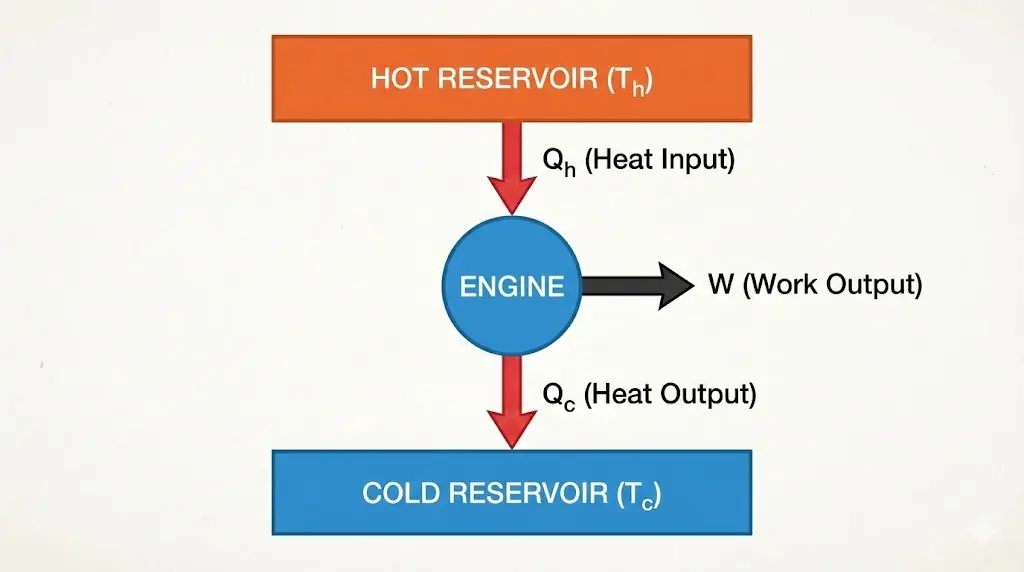
2. Efficiency of a Heat Engine
![]()
No heat engine can convert all absorbed heat into work. Some heat must always be rejected.
3. Carnot Engine and Maximum Efficiency
![]()
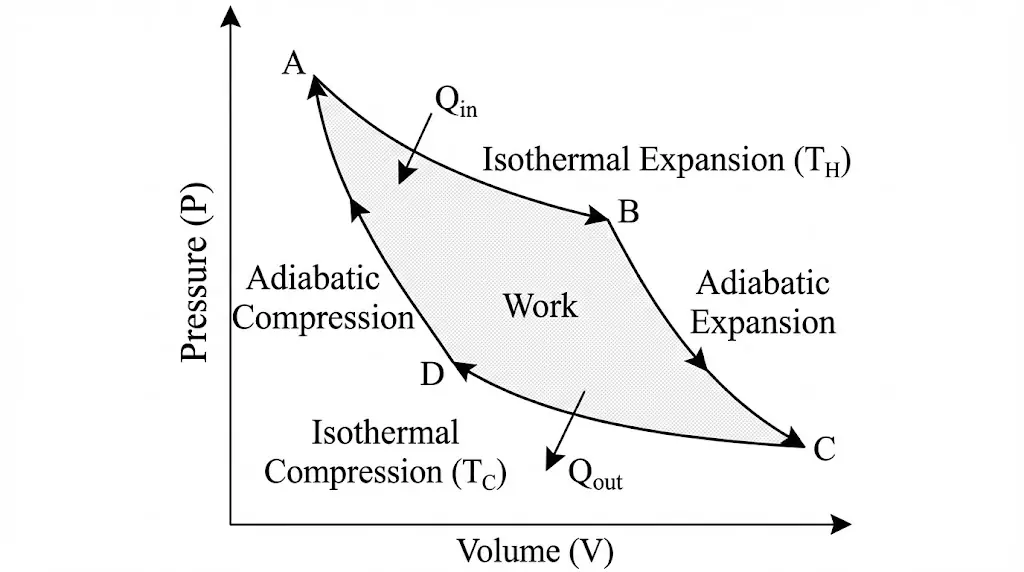
Carnot efficiency depends only on temperatures, not on the working substance.
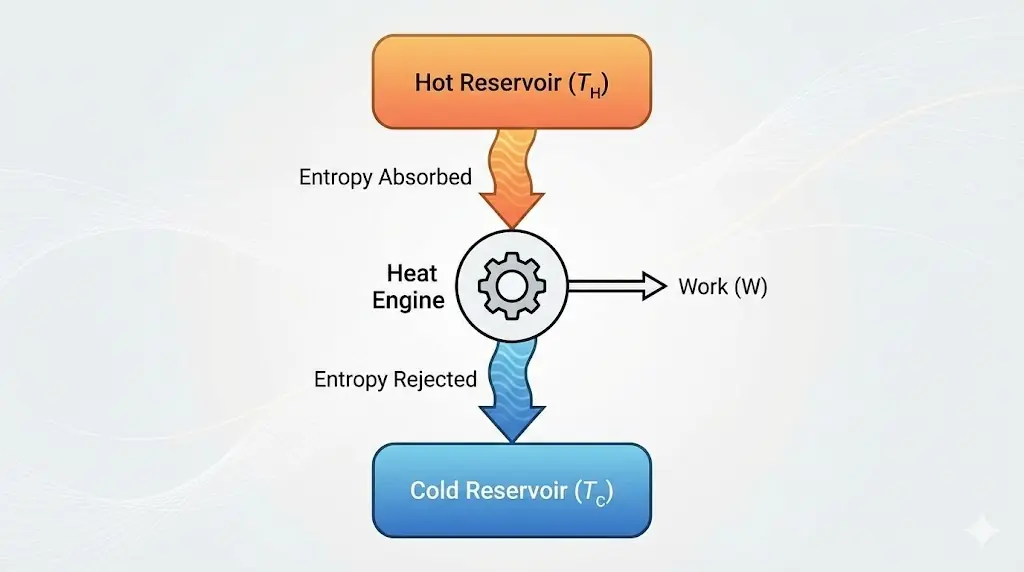
4. Entropy and Heat Engines
Even in ideal engines, entropy must be transferred to the cold reservoir.
5. Refrigerators and Heat Pumps
A refrigerator uses external work to extract heat from a cold region and reject it to a hotter environment.
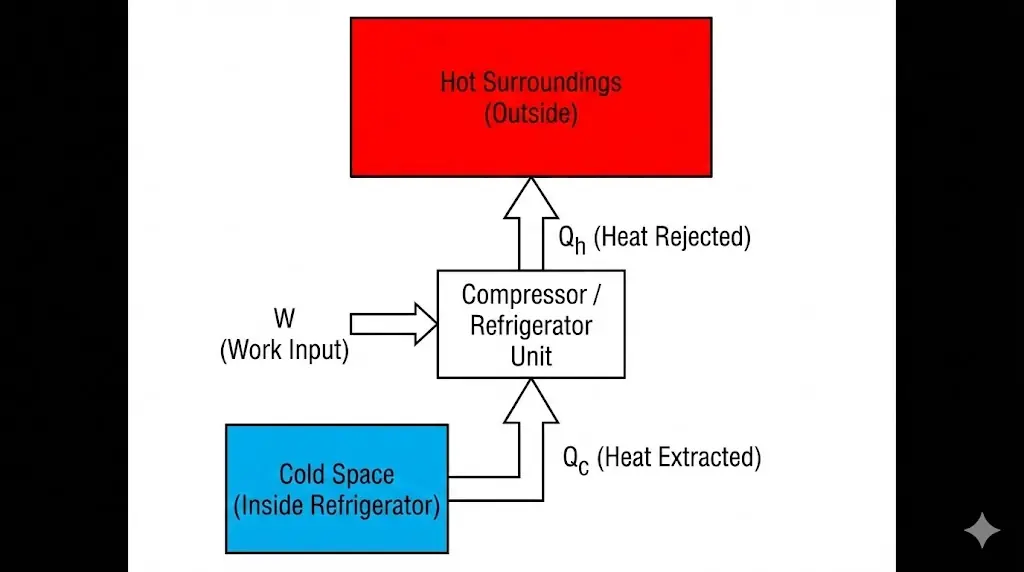
6. Coefficient of Performance (COP)
![]()
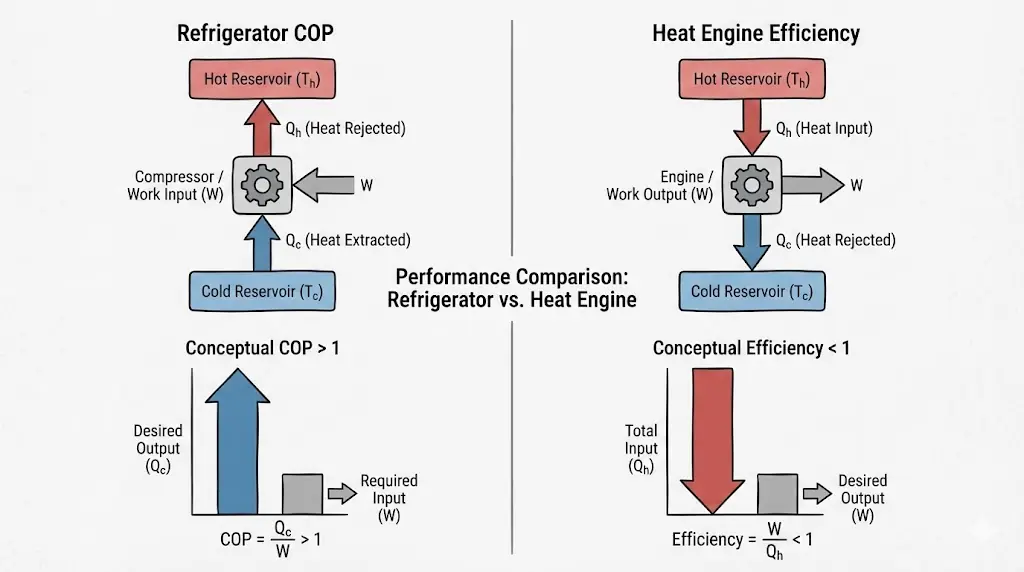
Refrigerators are judged by COP, not efficiency.
Practice Problems
Level 1 — Conceptual
Why must every heat engine reject heat to a cold reservoir?
Solution
Because entropy must be transferred to satisfy the second law.
Why is Carnot engine unattainable in practice?
Solution
It requires perfectly reversible processes.Level 2 — Numerical
An engine absorbs 500 J of heat and rejects 300 J. Find efficiency.
Solution
Efficiency = (500 − 300)/500 = 0.4.
A refrigerator removes 200 J of heat using 50 J of work. Find COP.
Solution
COP = 200/50 = 4.Level 3 — Advanced
Why does lowering cold reservoir temperature increase engine efficiency?
Solution
It increases the temperature difference, reducing entropy rejection.
Why is perpetual motion of second kind impossible?
