Advanced Physics → Modern Physics → Quantum Foundations

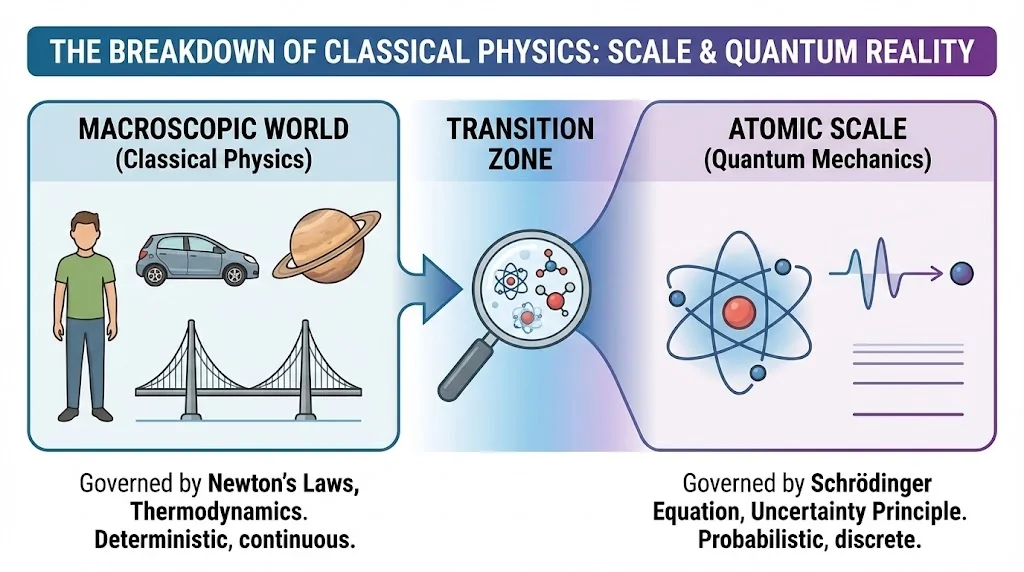
Quantum physics arose because classical physics could not explain atomic-scale phenomena.
1. Crisis of Classical Physics
By the end of the 19th century, classical physics seemed complete. However, several experiments produced results that could not be explained using Newtonian mechanics or classical electromagnetism.
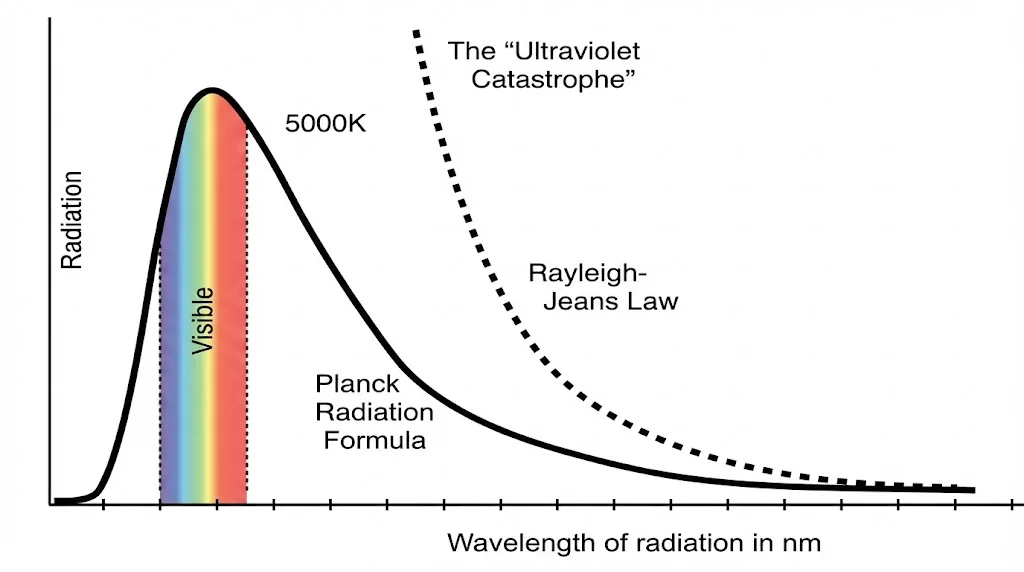
2. Blackbody Radiation
Classical theory predicted infinite energy emission at high frequencies, a result known as the ultraviolet catastrophe.
3. Photoelectric Effect
When light shines on a metal surface, electrons are emitted only if the light frequency exceeds a threshold value.
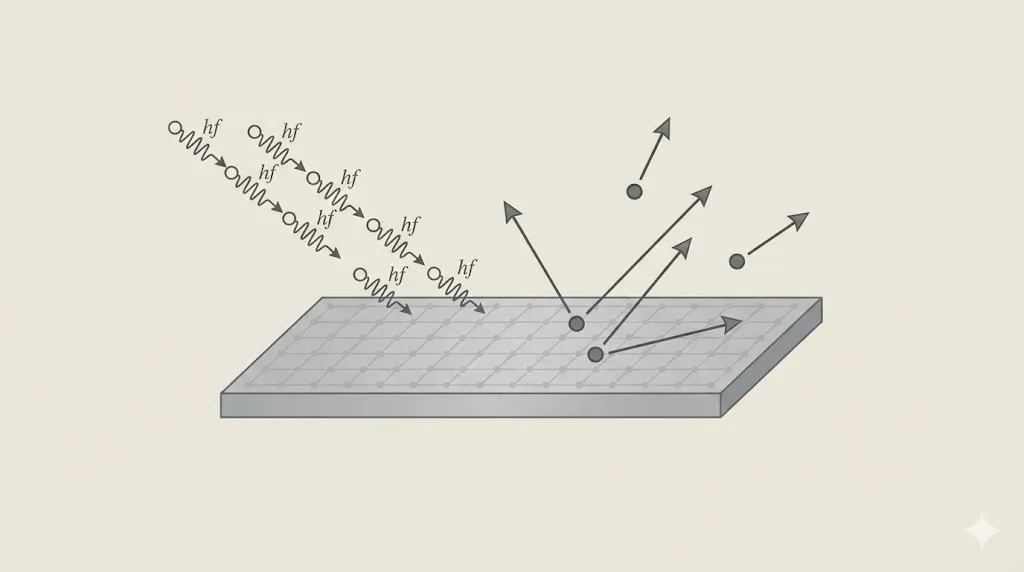
This showed that light behaves as particles called photons.
Einstein explained the effect using photons with energy ![]() , giving the relation
for maximum kinetic energy
, giving the relation
for maximum kinetic energy ![]() , where
, where ![]() is the work function.
is the work function.
4. Wave–Particle Duality
Light behaves as both a wave and a particle. Surprisingly, matter such as electrons also exhibits wave-like behavior.

5. de Broglie Hypothesis
Louis de Broglie proposed that every particle has an associated wavelength.
![]()

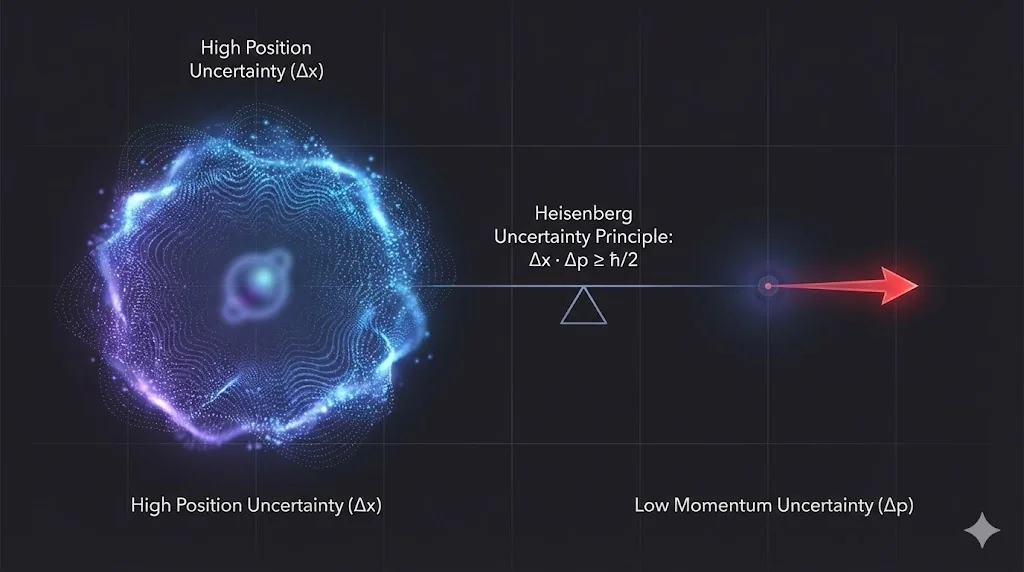
6. Heisenberg Uncertainty Principle
It is fundamentally impossible to know both the exact position and momentum
of a particle simultaneously; their uncertainties obey
![]() .
.
7. Breakdown of Classical Concepts
At atomic scales, determinism is replaced by probability. Physical quantities are described statistically rather than exactly.