Advanced Physics → Modern Physics → Atomic Structure
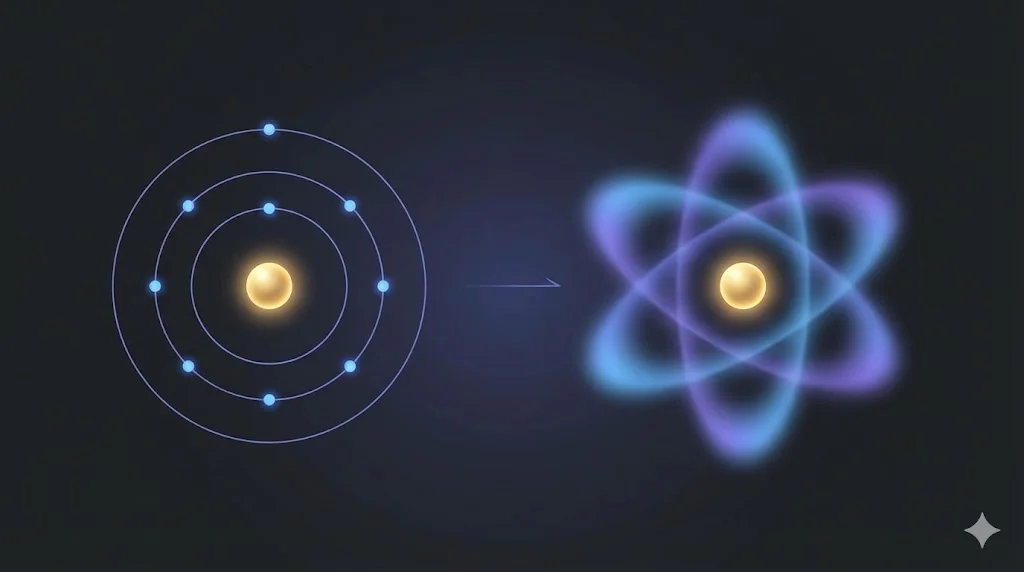
Why do atoms remain stable, emit discrete spectra, and obey quantized energy rules?
1. Why Atomic Structure Is a Fundamental Problem
Atoms are the building blocks of matter, yet their stability cannot be explained using classical mechanics or electrodynamics.
Understanding atomic structure forced physics to abandon classical continuity and introduce quantization.
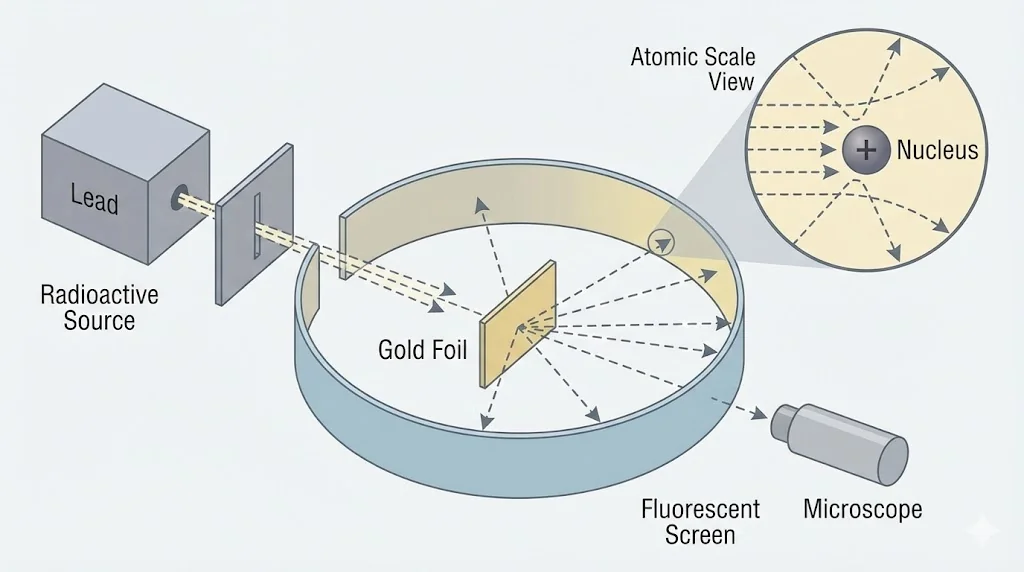
2. Experimental Clue: Rutherford Scattering
Scattering experiments showed that atoms contain a tiny, dense, positively charged nucleus.
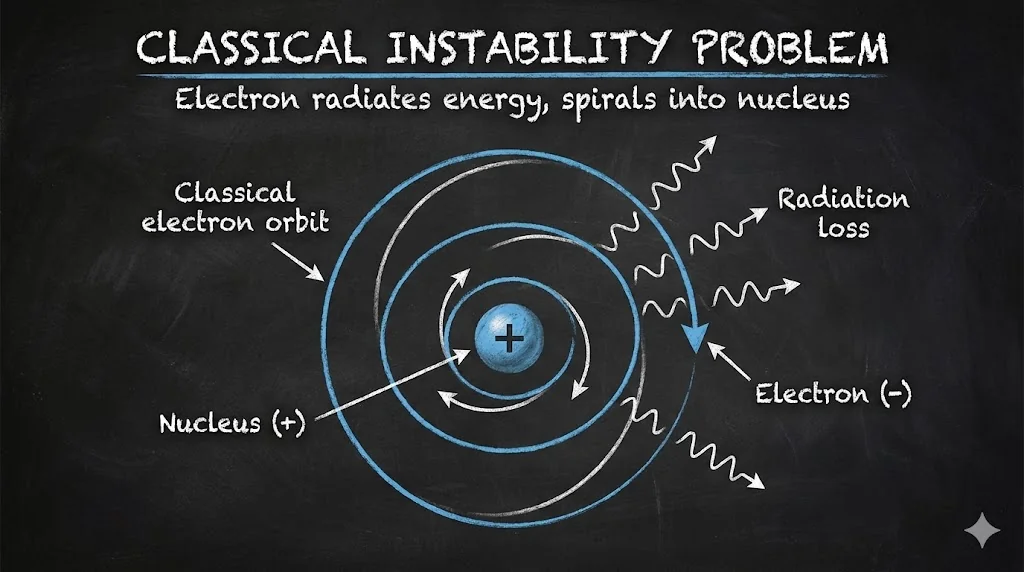
3. Failure of Classical Atomic Models
According to classical physics, an accelerating electron must radiate energy and spiral into the nucleus.
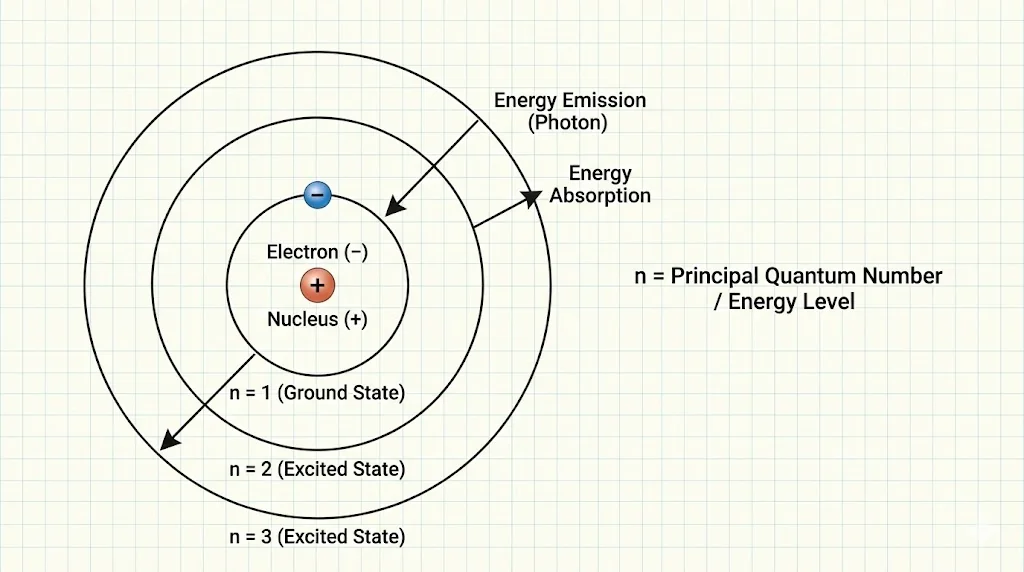
4. Bohr’s Quantization Hypothesis
Bohr proposed that electrons can occupy only certain allowed orbits without radiating energy.
![]()
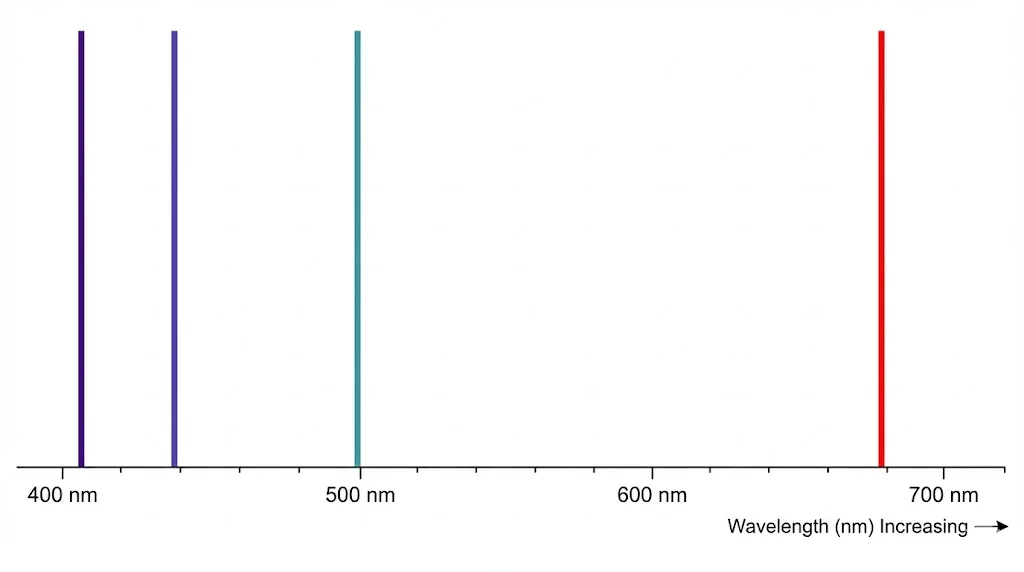
5. Spectral Evidence
Atoms emit and absorb light at specific wavelengths, corresponding to transitions between energy levels. The visible hydrogen lines form the Balmer series (n=2 transitions).
6. Wave–Particle Duality
The electron exhibits both particle and wave behavior. Standing wave conditions explain allowed orbits.

7. Quantum View of the Atom
Modern quantum mechanics replaces orbits with probability distributions.

