Advanced Physics → Modern Physics → Semiconductors & Solid State
Central question
How does arranging atoms in a crystal radically change electrical behavior?
How does arranging atoms in a crystal radically change electrical behavior?
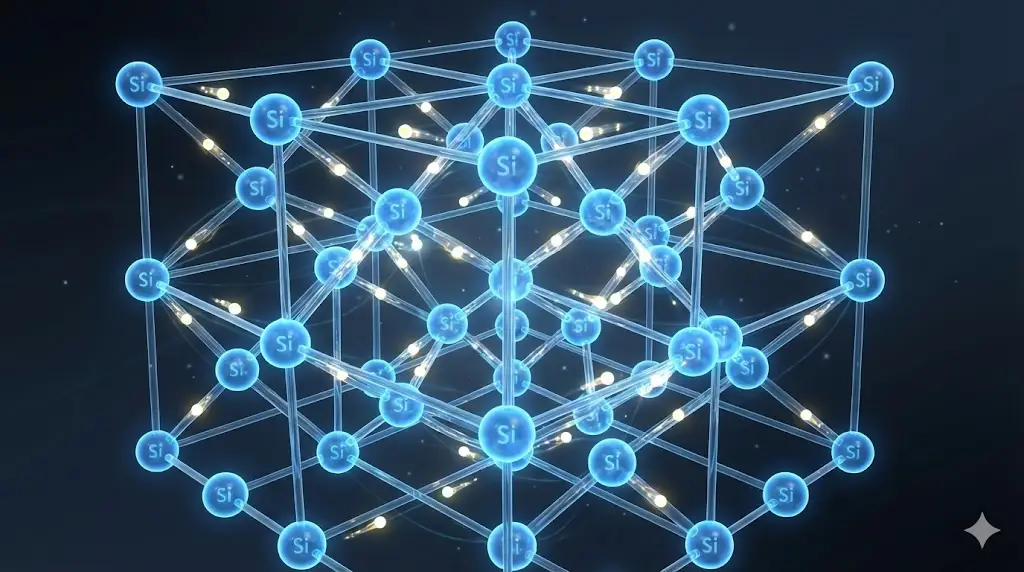
1. Solids as Collective Systems
In solids, atoms are not isolated. Their electrons interact strongly with neighboring atoms, producing properties that cannot be understood at the single-atom level.
2. Energy Bands in Solids
When atoms form a crystal, discrete atomic energy levels broaden into bands.
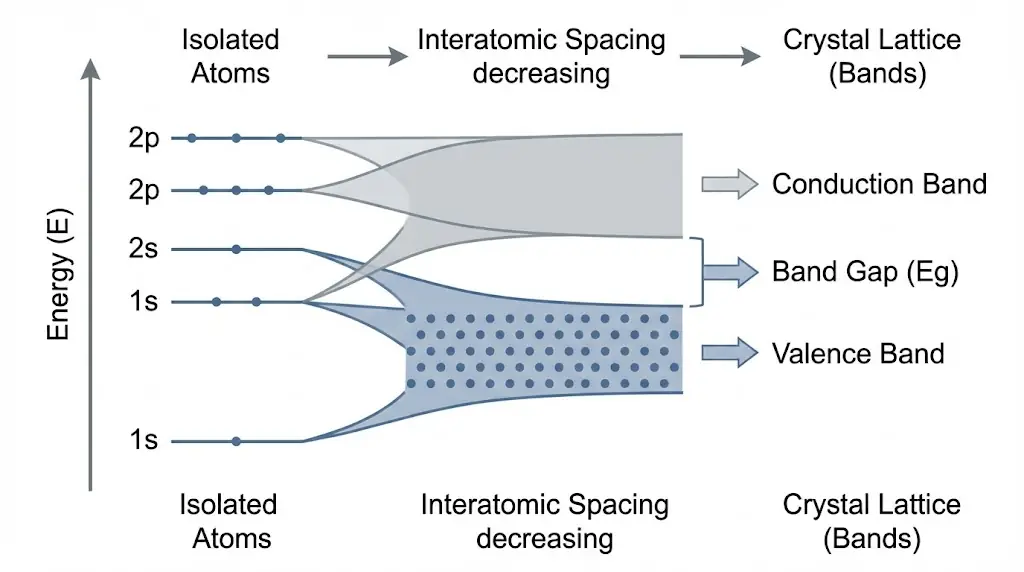
Electrical behavior depends on how electrons populate these bands.
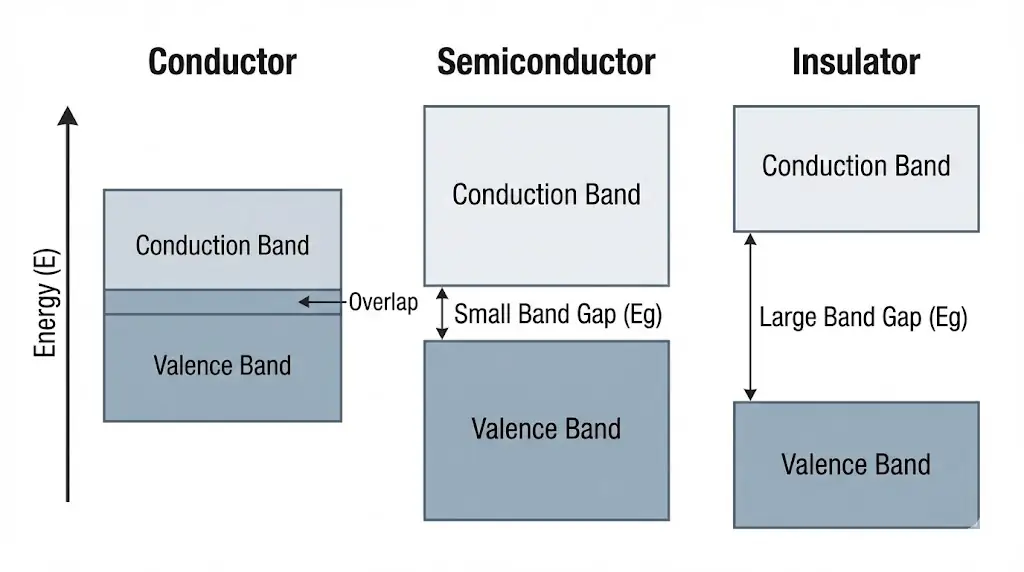
3. Conductors, Insulators, and Semiconductors
Materials differ based on the energy gap between bands.
4. Intrinsic Semiconductors
In pure semiconductors, thermal energy can excite electrons from the valence band to the conduction band.
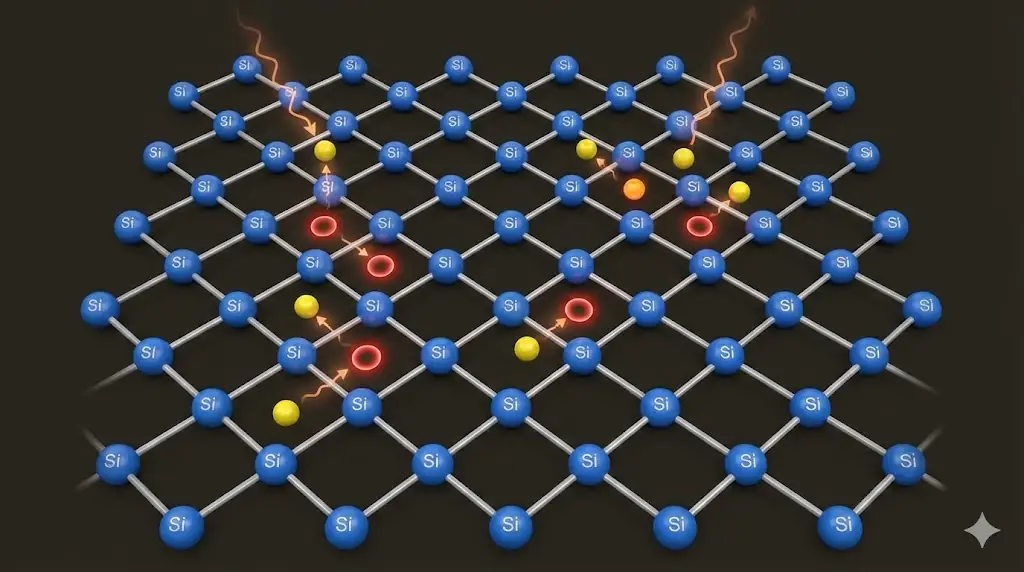
5. Doping and Charge Control
Adding small amounts of impurities dramatically alters conductivity.
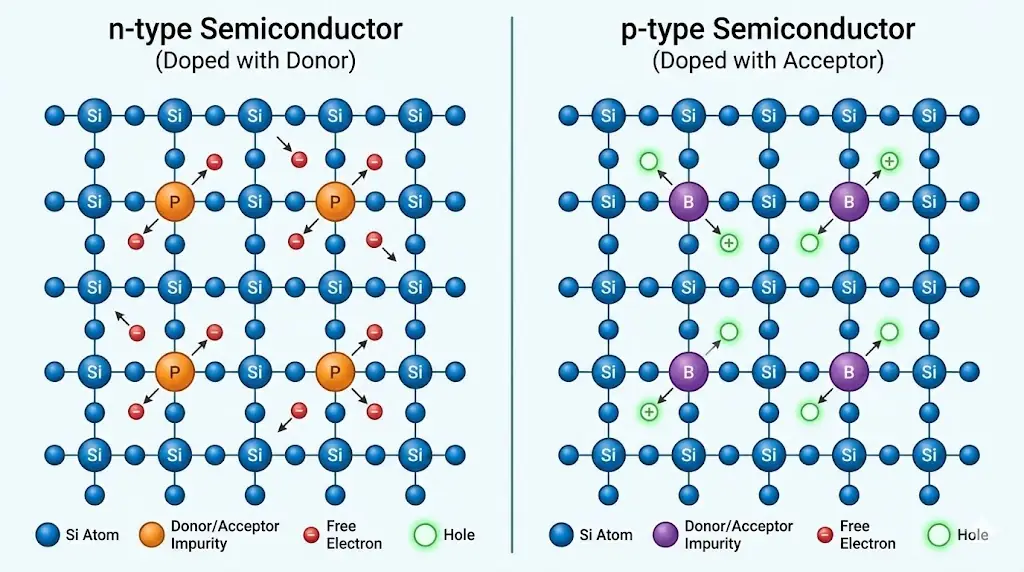
6. p–n Junction: A New Physical Entity
Joining p-type and n-type regions creates a depletion region and an internal electric field.
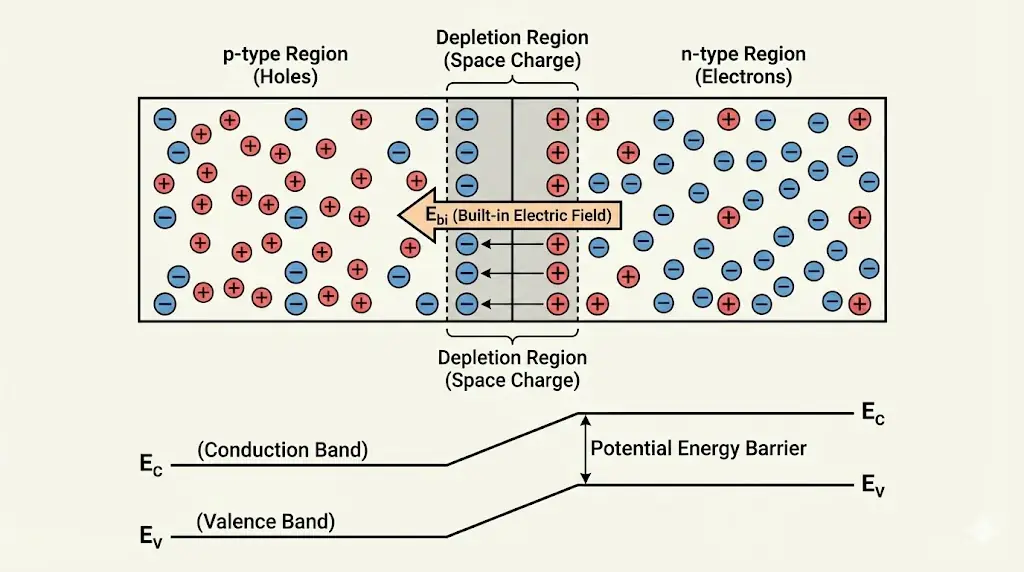
Semiconductor devices work by controlling carrier motion, not creating energy.
7. Why Semiconductors Matter
Semiconductors underpin nearly all modern technology.

Conceptual Problems
Why do energy bands not exist in isolated atoms?
Answer
Bands arise from interactions between large numbers of atoms.
Why can holes behave like positive charge carriers?
Answer
Their motion corresponds to missing electrons moving through the lattice.
Why is doping so effective despite tiny impurity concentrations?
