Advanced Physics → Advanced Thermal Physics → Kinetic Theory of Gases
• Gas pressure arises from molecular collisions
• Temperature measures average kinetic energy
• Calculus and statistics bridge micro and macro physics
This page is for learners who know basic thermodynamics and want a microscopic, calculus-based view of gases rather than equation memorization.
1. Why Kinetic Theory Matters
Thermodynamics describes gases using macroscopic variables such as pressure, volume, and temperature.
Kinetic theory explains these quantities by modeling the microscopic motion of a very large number of particles.
2. Microscopic vs Macroscopic Descriptions
At the microscopic level, a gas consists of molecules in constant random motion.
At the macroscopic level, we observe smooth quantities such as pressure and temperature, which represent statistical averages over trillions of particles.
3. Assumptions of the Ideal Gas Model
- Molecules are point particles
- Intermolecular forces are negligible except during collisions
- Collisions are perfectly elastic
- Motion is random and isotropic
These assumptions allow a tractable mathematical description.
4. Pressure from Molecular Collisions
Consider molecules colliding elastically with the walls of a container. Each collision transfers momentum to the wall.
![]()
Summing momentum transfer over many collisions leads to pressure.
![]()
5. Root-Mean-Square (RMS) Speed
Because molecular speeds vary, an average speed must be defined statistically.
![]()
Using kinetic theory:
![]()
![]()
6. Temperature and Kinetic Energy
Temperature is a measure of the average translational kinetic energy of gas molecules.
Higher temperature corresponds to higher molecular speeds.
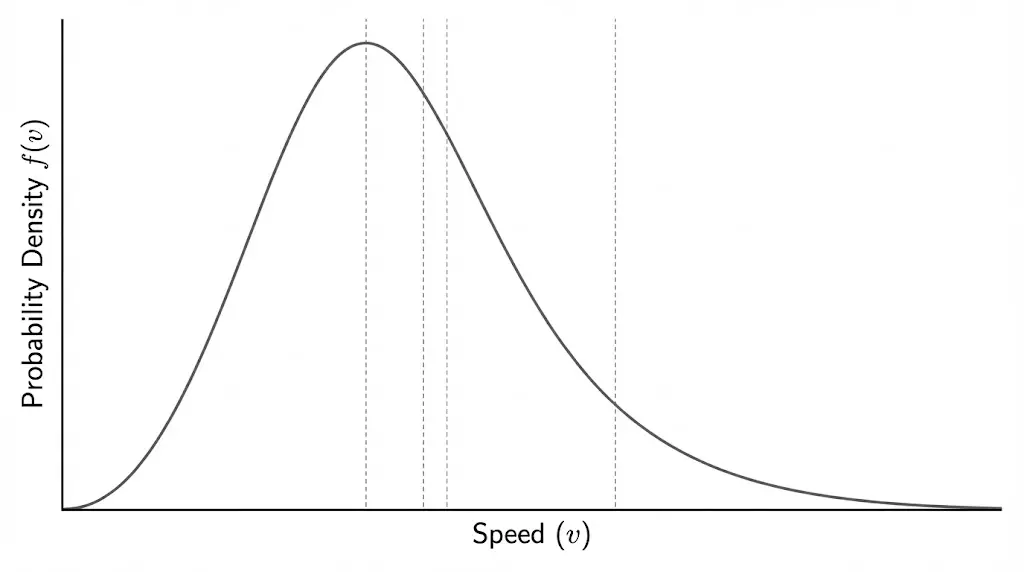
7. Maxwell–Boltzmann Speed Distribution
Molecular speeds follow a statistical distribution rather than a single value.
![]()
8. Limitations of Kinetic Theory
- Fails at very high densities
- Breaks down at low temperatures
- Ignores intermolecular attractions
These limitations motivate real-gas and statistical mechanics models.
