Advanced Physics → Modern Physics → Nuclear Physics
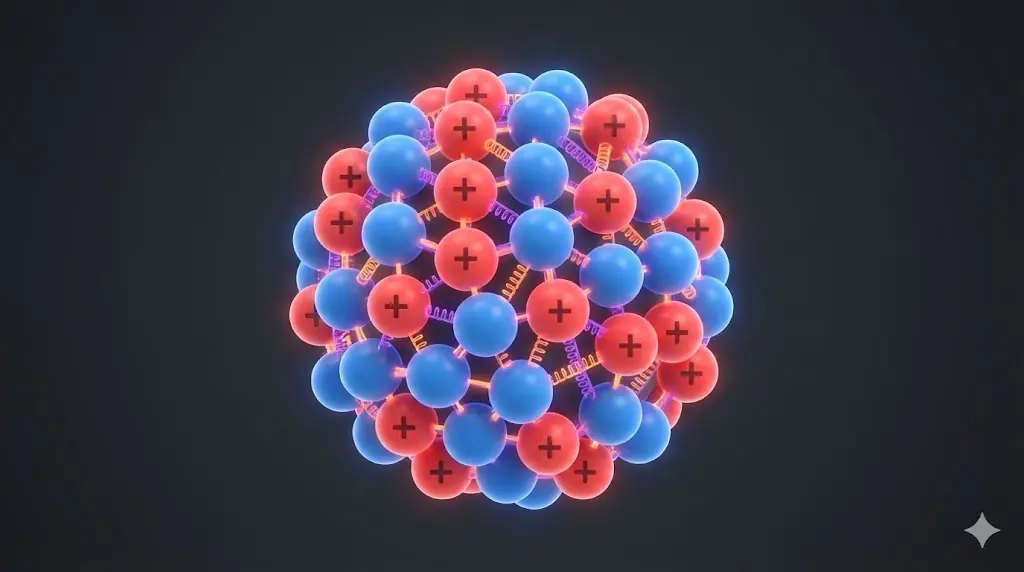
Core question
Why do atomic nuclei exist at all despite intense electrostatic repulsion?
Why do atomic nuclei exist at all despite intense electrostatic repulsion?
1. The Nuclear Scale
The nucleus occupies only a tiny fraction of atomic volume yet contains nearly all the atom’s mass.
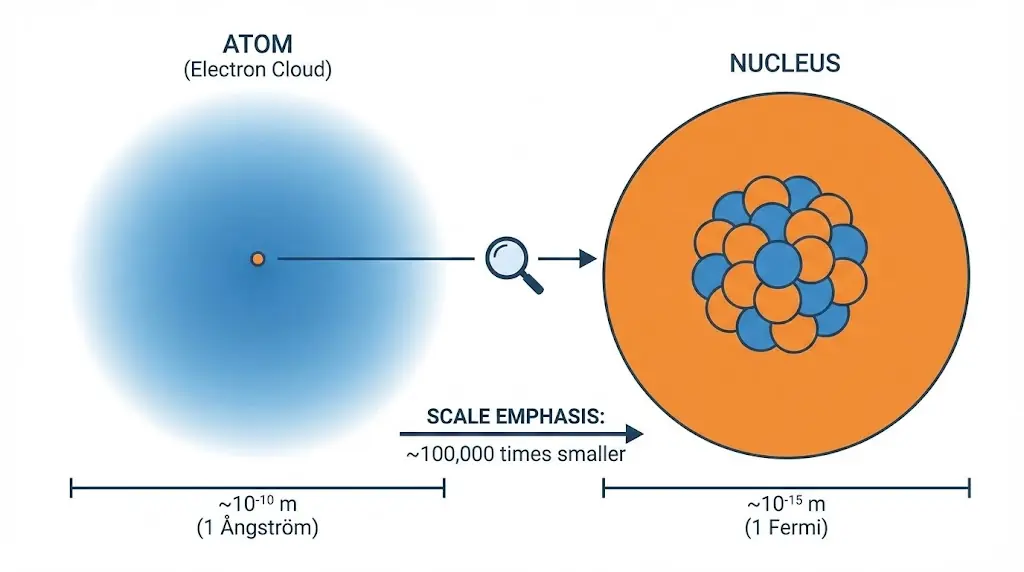
2. The Strong Nuclear Force
Protons repel each other electrically. A new force is required to bind the nucleus.
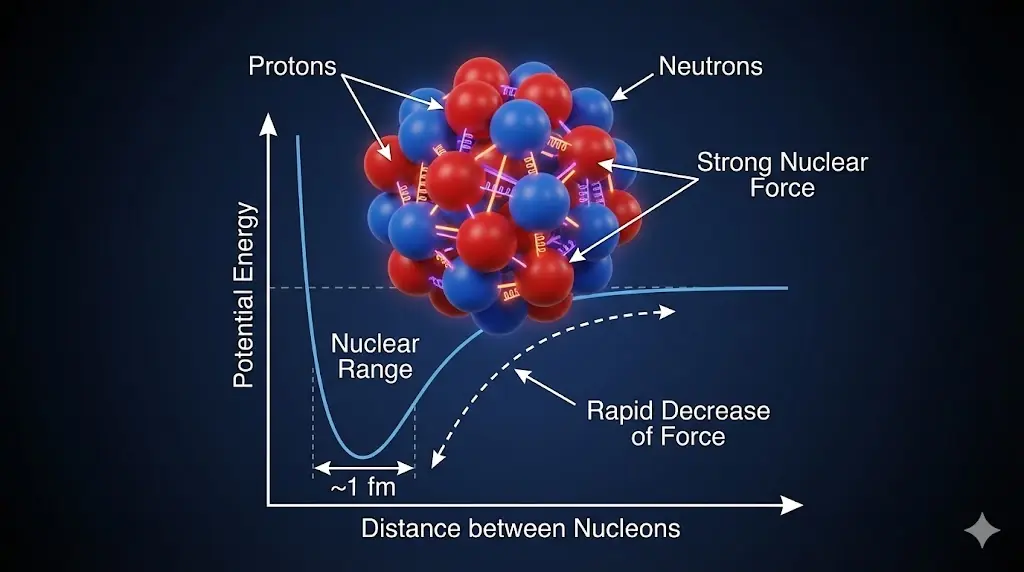
The strong nuclear force overwhelms electrostatic repulsion inside nuclei.
3. Nuclear Binding Energy
Binding energy measures how strongly nucleons are held together.
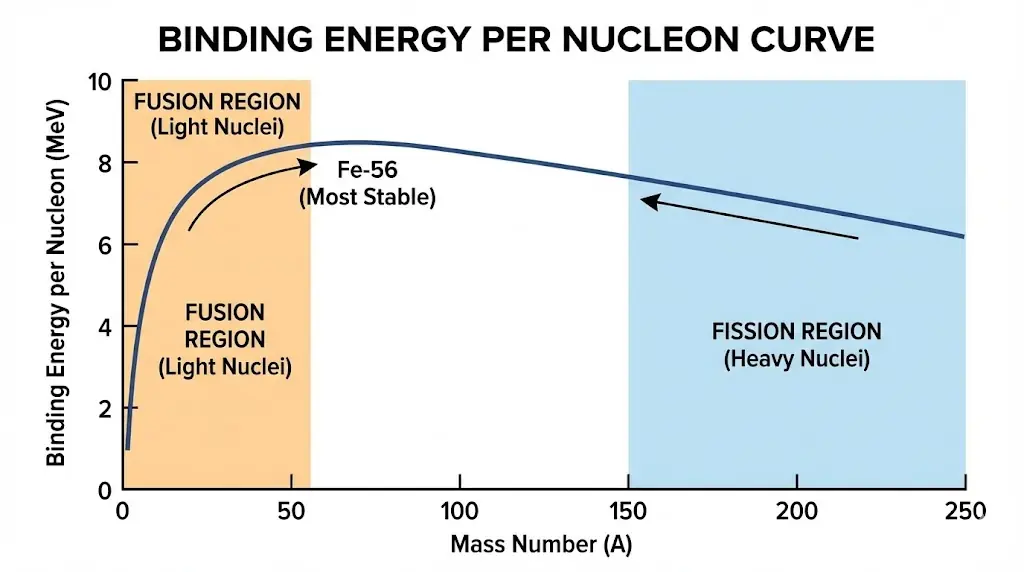
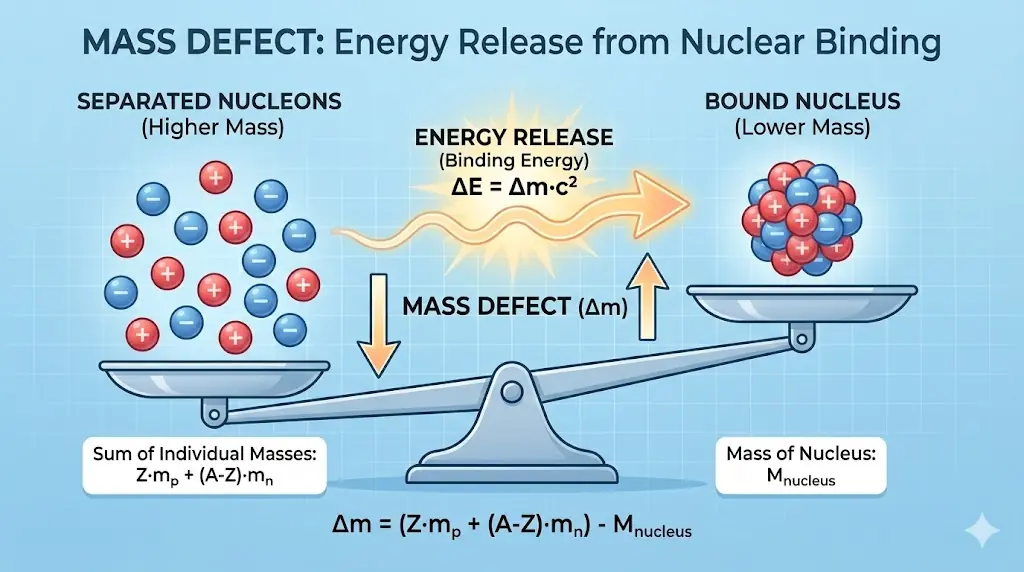
4. Mass Defect and Energy
Bound nuclei have less mass than their separated nucleons. The missing mass appears as binding energy.
\[
E = \Delta m c^2
\]
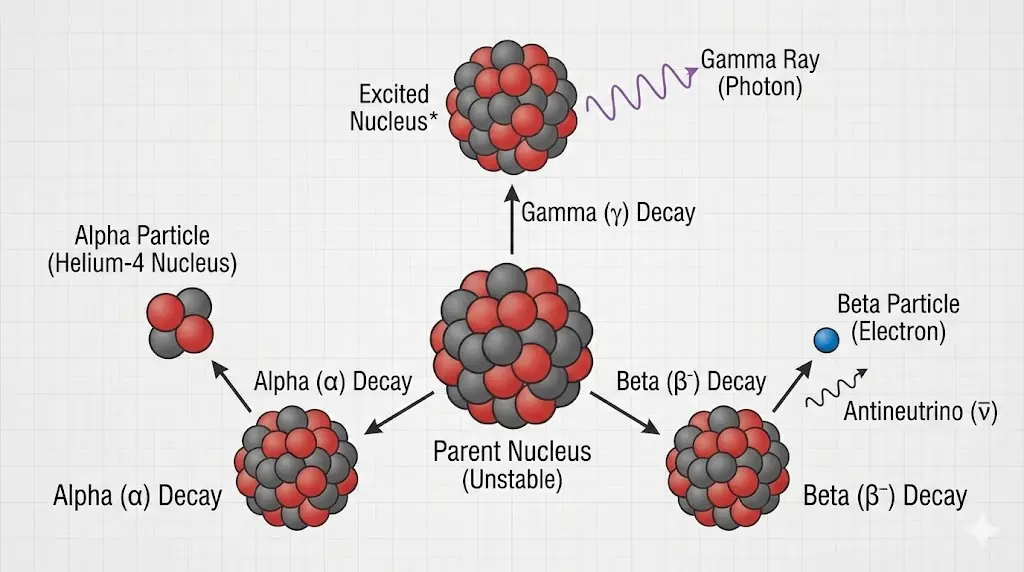
5. Nuclear Stability and Radioactivity
Some nuclei are unstable and decay spontaneously, transforming into more stable configurations.
6. Fission and Fusion
Energy can be released by splitting heavy nuclei or combining light nuclei.
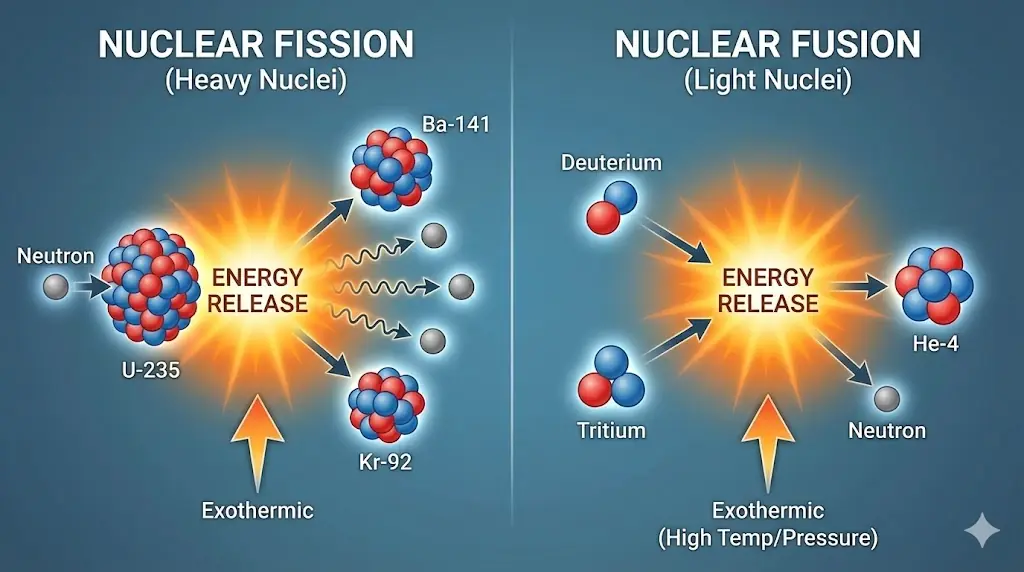
Nuclear energy arises from changes in binding energy, not chemical bonds.
7. Why Nuclear Physics Matters
- Stellar energy generation
- Radioactive dating
- Nuclear medicine
- Fundamental particle research
Conceptual Problems
Why does electrostatic repulsion not destroy the nucleus?
Answer
The strong nuclear force dominates at very short distances.
Why is iron among the most stable nuclei?
Answer
It has maximum binding energy per nucleon.
Why does mass decrease when a nucleus forms?
