Thermal Properties of Matter
NCERT Chapter 10 • Heat, Expansion, Calorimetry & Heat Transfer

1. Temperature and Scales
Temperature is a relative measure of hotness or coldness. Heat is the form of energy transferred between two systems (or a system and its surroundings) by virtue of temperature difference.
1.1 Temperature Scales
| Scale | Freezing Point | Boiling Point | Conversion |
|---|---|---|---|
| Celsius ( | 0 | 100 | – |
| Fahrenheit ( | 32 | 212 | |
| Kelvin (K) | 273.15 | 373.15 |
1.2 Ideal Gas Equation
Combining Boyle’s Law (![]() at constant
at constant ![]() ) and Charles’ Law (
) and Charles’ Law (![]() at constant
at constant ![]() ), we get:
), we get:
where ![]() = number of moles,
= number of moles, ![]() J/mol·K (universal gas constant).
J/mol·K (universal gas constant).
1.3 Absolute Zero
Experiments with low-density gases show that at constant volume, ![]() . Extrapolating to
. Extrapolating to ![]() gives
gives ![]() C, defined as absolute zero (0 K).
C, defined as absolute zero (0 K).
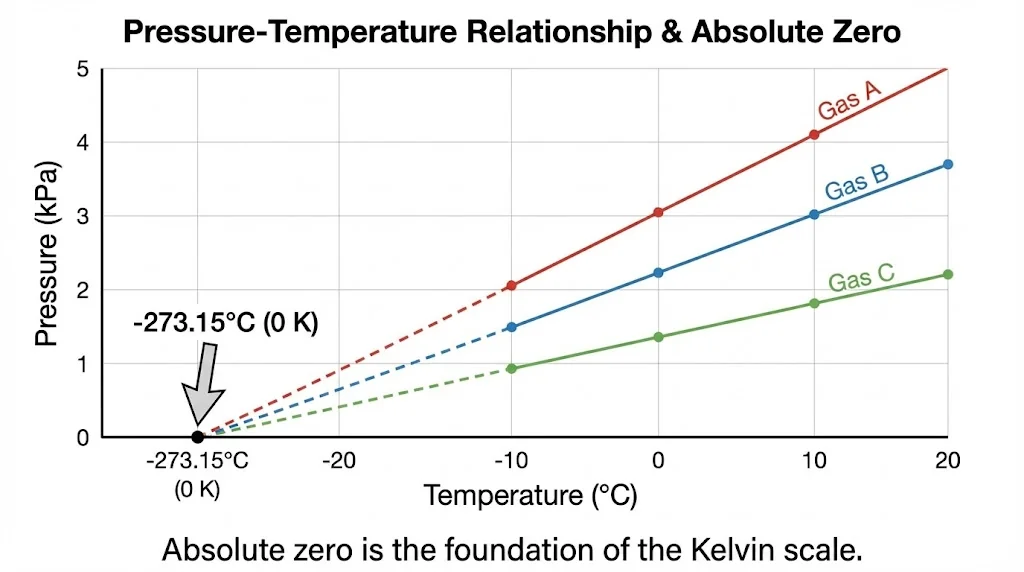
1.4 Triple Point of Water
The triple point is the unique temperature and pressure where water coexists as solid, liquid, and vapor in equilibrium:
- Temperature = 273.16 K (0.01°C)
- Pressure = 6.11 × 10⁻³ Pa
This is used to define the Kelvin scale because it’s a reproducible fixed point.
2. Thermal Expansion
Most substances expand on heating and contract on cooling.
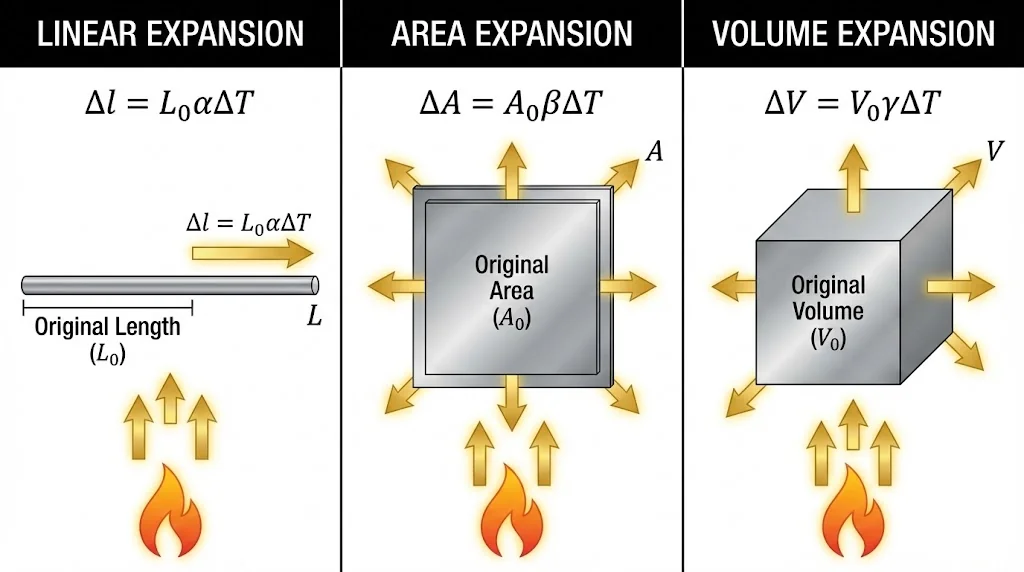
 ).
).2.1 Types of Expansion
- Linear Expansion (
 ):
):  (1D: Length)
(1D: Length) - Area Expansion (
 ):
):  (2D: Area)
(2D: Area) - Volume Expansion (
 ):
):  (3D: Volume)
(3D: Volume)
Relation: ![]() . For solids,
. For solids, ![]() .
.
2.2 Thermal Stress
When a rod is rigidly fixed and heated, it cannot expand. This induces compressive stress:
where ![]() = Young’s modulus.
= Young’s modulus.
Water contracts on heating from 0![]() C to 4
C to 4![]() C. Its density is maximum at 4
C. Its density is maximum at 4![]() C. This property allows aquatic life to survive in frozen lakes.
C. This property allows aquatic life to survive in frozen lakes.
Show Answer
The tape expands on the hot day, so its markings are farther apart. It measures less than the actual length.Expansion factor =
True Length = Measured Length
3. Calorimetry
Calorimetry implies measurement of heat. Principle: Heat Lost = Heat Gained.
3.1 Specific Heat Capacity ( or
or  )
)
The amount of heat required to raise the temperature of unit mass of a substance by 1![]() C.
C.
For water, ![]() J kg
J kg![]() K
K![]() (very high). This makes water an excellent coolant.
(very high). This makes water an excellent coolant.
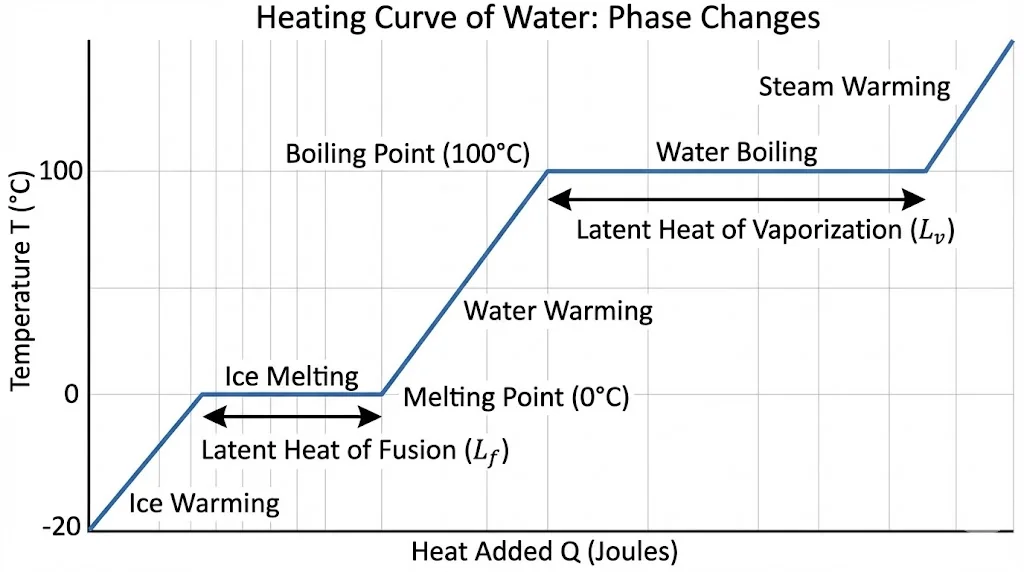
3.2 Change of State & Latent Heat ( )
)
During a phase change (Melting/Boiling), temperature remains constant. Heat supplied is used to break bonds.
- Latent Heat of Fusion (
 ): Solid
): Solid  Liquid. (Ice:
Liquid. (Ice:  J/kg)
J/kg) - Latent Heat of Vaporisation (
 ): Liquid
): Liquid  Gas. (Water:
Gas. (Water:  J/kg)
J/kg)
3.3 Molar Specific Heat for Gases
For gases, heat capacity depends on process:
 : At constant volume (no work done).
: At constant volume (no work done). : At constant pressure (work done in expansion).
: At constant pressure (work done in expansion).
Always ![]() because extra heat is needed for work. For monatomic gases,
because extra heat is needed for work. For monatomic gases, ![]() .
.
3.4 Regelation
When pressure is applied to ice, its melting point lowers. A weighted wire can pass through an ice block because ice melts under the wire and refreezes above it. This is called regelation.
(
Show Answer
1. Ice (-12 to 0):2. Melt Ice (0 to 0):
3. Water (0 to 100):
4. Boil Water (100 to 100):
Total
4. Heat Transfer
Heat flows from high temperature to low temperature via three modes:
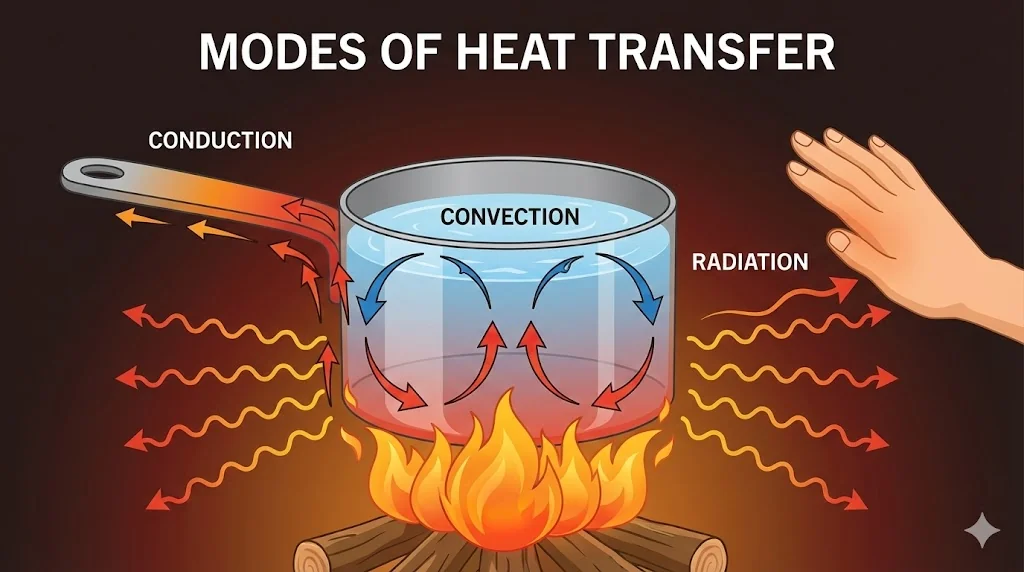
4.1 Conduction
Heat transfer by molecular collision without bulk motion of matter (mostly solids). Rate of heat flow (![]() ):
):
Where ![]() is Thermal Conductivity.
is Thermal Conductivity. ![]() .
.
4.2 Thermal Conductivity Table
| Material | K (W/m·K) |
|---|---|
| Silver | 406 |
| Copper | 385 |
| Water | 0.8 |
| Air | 0.024 |
4.3 Series Combination of Rods
For two rods in series (same heat current ![]() ):
):

4.4 Convection
Heat transfer by actual motion of fluid particles (liquids & gases). Examples: Sea breeze, trade winds.
4.5 Radiation
Heat transfer via electromagnetic waves (infrared). Needs no medium. Example: Sunlight.
4.6 Dewar Flask (Thermos)
A thermos minimizes heat transfer by:
- Conduction: Vacuum between double walls.
- Convection: No air in vacuum.
- Radiation: Silvered walls reflect heat.
5. Newton’s Law of Cooling
The rate of loss of heat (![]() ) of a body is directly proportional to the temperature difference between the body and its surroundings (
) of a body is directly proportional to the temperature difference between the body and its surroundings (![]() ), provided
), provided ![]() is small.
is small.
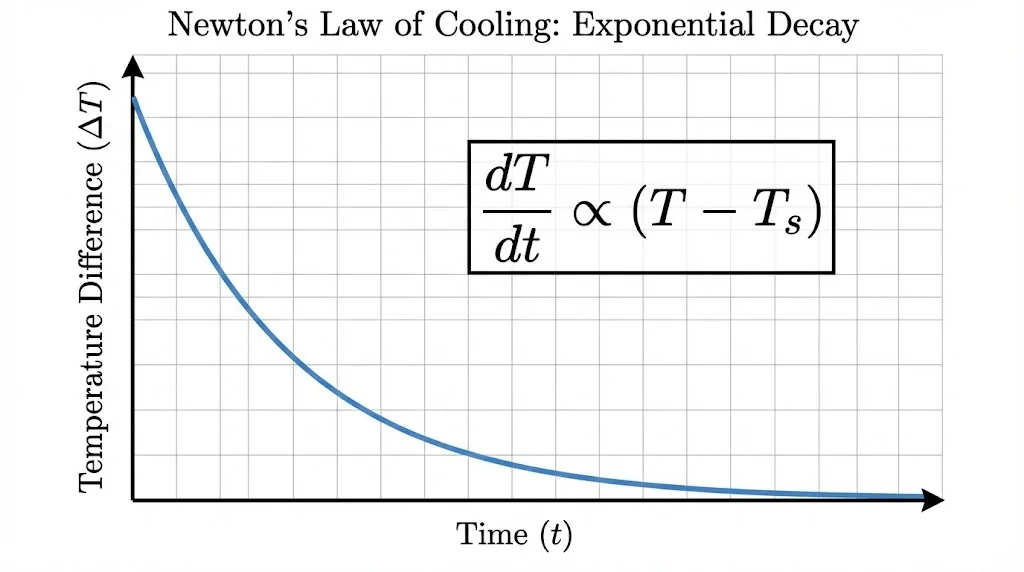
Show Answer
Using Avg Rate form:Case 1:
Case 2:
6. Blackbody Radiation Laws
Stefan-Boltzmann Law
Energy radiated per second per unit area by a blackbody is proportional to the fourth power of its absolute temperature.
![]() (Stefan’s Constant).
(Stefan’s Constant).
6.1 Emissivity
A perfect blackbody has emissivity ![]() . Real bodies have
. Real bodies have ![]() . The net power radiated is:
. The net power radiated is:
Wien’s Displacement Law
The wavelength (![]() ) corresponding to maximum emission intensity is inversely proportional to absolute temperature.
) corresponding to maximum emission intensity is inversely proportional to absolute temperature.
This explains why hotter stars appear blue (lower ![]() ) and cooler stars appear red (higher
) and cooler stars appear red (higher ![]() ).
).
