Thermodynamics
NCERT Chapter 11 • Laws of Thermodynamics, Processes, Heat Engines & Carnot Cycle
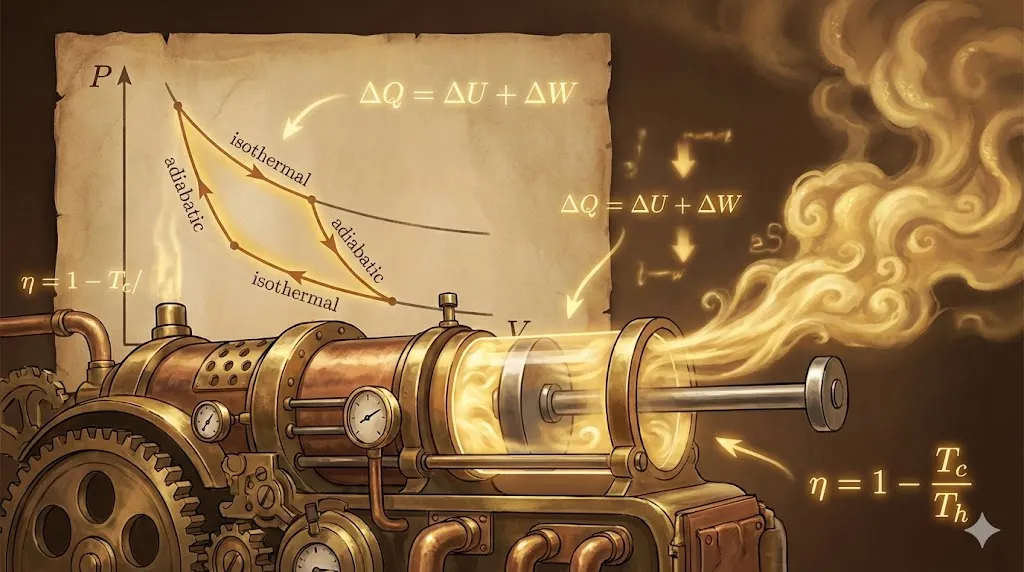
1. Thermal Equilibrium & Zeroth Law
Two systems are in thermal equilibrium if they have the same temperature and no net heat flows between them when connected by a conducting (diathermic) wall.
The Zeroth Law of Thermodynamics states:
“If two systems A and B are separately in thermal equilibrium with a third system C, then A and B are also in thermal equilibrium with each other.”
This law justifies the concept of temperature as a physical quantity that determines the direction of heat flow.
2. First Law of Thermodynamics
This is the principle of conservation of energy applied to thermodynamic systems.
- ΔQ: Heat supplied to the system (+ve if absorbed).
- ΔU: Change in internal energy (depends only on initial and final states).
- ΔW: Work done by the system (+ve if system expands).
Work Done by a Gas
For a gas expanding from volume V₁ to V₂:
![]()
Geometrically, this is the area under the P–V curve.
Show Answer
Using 1st Law: ΔQ = ΔU + ΔW.200 = ΔU + 50 ⇒ ΔU = 150 J.
3. Specific Heat Capacity
When heat ΔQ is supplied to a substance, its temperature changes by ΔT. We define:
- Specific heat capacity (s): Heat required to raise the temperature of 1 kg of a substance by 1 K.
 [Unit: J kg⁻¹ K⁻¹]
[Unit: J kg⁻¹ K⁻¹] - Molar specific heat capacity (C): Heat required to raise the temperature of 1 mole by 1 K.
 [Unit: J mol⁻¹ K⁻¹]
[Unit: J mol⁻¹ K⁻¹]
Calorie Definition
Historically, 1 calorie was defined as the heat needed to raise 1 g of water from 14.5°C to 15.5°C.
Now, 1 cal = 4.186 J. In SI units, we use joules exclusively.
Specific Heat of Solids
Using the law of equipartition of energy, each atom in a solid has 6 degrees of freedom (3 kinetic + 3 potential). Thus, average energy per mole is:
This matches experimental values for most solids at room temperature (see table below).
| Substance | Specific Heat (J kg⁻¹ K⁻¹) | Molar Heat (J mol⁻¹ K⁻¹) |
|---|---|---|
| Aluminium | 900 | 24.4 |
| Copper | 385 | 24.5 |
| Iron | 470 | 26.0 |
| Lead | 130 | 27.0 |
Relation for Ideal Gases: 
For 1 mole of ideal gas:
- At constant volume:

- At constant pressure:

Hence,
where γ = Cp/Cv is used in adiabatic processes.
Derivation: Cp − Cv = R for Ideal Gas 3 Marks • CBSE Important
🔍 Show Derivation
At constant volume, no work is done (ΔV = 0), so from First Law:
ΔQ = ΔU + PΔV = ΔU + 0 = ΔU
Molar specific heat at constant volume:
At constant pressure, work done by gas:
From First Law:
Molar specific heat at constant pressure:
Since internal energy of ideal gas depends only on temperature:
For 1 mole:
At constant pressure:
→
This relation holds only for ideal gases. For real gases, ![]() due to intermolecular forces.
due to intermolecular forces.
4. Thermodynamic State Variables & Equation of State
An equilibrium state of a system is fully described by **state variables**: pressure (P), volume (V), temperature (T), internal energy (U), and composition.
Extensive vs. Intensive Variables
- Extensive: Depend on system size (e.g., mass, volume, internal energy). Halved if system is split.
- Intensive: Independent of size (e.g., pressure, temperature, density). Unchanged if system is split.
In the First Law: ![]() , all terms are extensive. (P is intensive, ΔV is extensive ⇒ PΔV is extensive.)
, all terms are extensive. (P is intensive, ΔV is extensive ⇒ PΔV is extensive.)
Non-Equilibrium States
State variables are undefined for non-equilibrium states, such as:
- Free expansion into vacuum (no uniform P or T)
- Explosive chemical reactions (e.g., petrol-air mixture ignition)
Only after equilibrium is restored can we assign P, V, T.
5. Thermodynamic Processes
A process describes how a system changes from one equilibrium state to another.
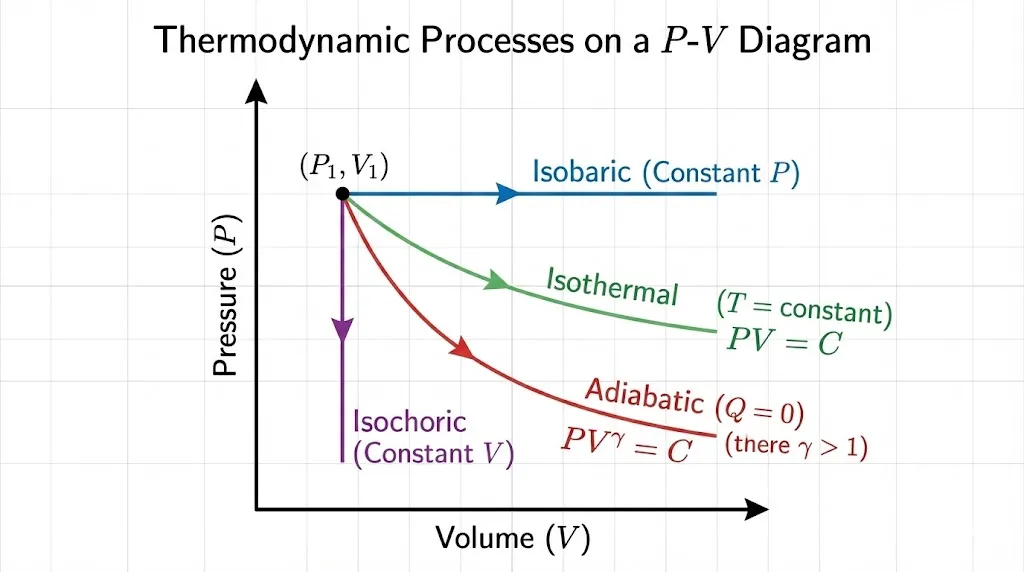
| Process | Constraint | Relation | Work Done (W) |
|---|---|---|---|
| Isothermal | Constant Temperature (ΔT = 0) | PV = constant | nRT ln(V₂/V₁) |
| Adiabatic | No Heat Exchange (ΔQ = 0) | PVγ = constant | (P₁V₁ − P₂V₂)/(γ − 1) |
| Isobaric | Constant Pressure (ΔP = 0) | V/T = constant | P(V₂ − V₁) |
| Isochoric | Constant Volume (ΔV = 0) | P/T = constant | 0 |
Derivation of Work Done in Thermodynamic Processes 4 Marks • JEE Focus
🔍 Show Full Derivation
dW = Force × displacement = (P × A) × dx = P × (A dx) = P dV
Total work from
(Geometrically: area under P–V curve)
From ideal gas law: PV = nRT ⇒ P = nRT / V
→
→
→
For adiabatic process:
→
![Rendered by QuickLaTeX.com W = \int^{V_2}_{V_1}K V^{-\gamma} dV = K \left[\dfrac{V^{1-\gamma}}{1-\gamma}\right]^{V_2}_{V_1}](https://i0.wp.com/physicsqanda.com/wp-content/ql-cache/quicklatex.com-6c789a31249595b2cc50785fa9660817_l3.png?resize=267%2C50&ssl=1)
→
Using
→
→
→
→
Since
→
Work depends on the path taken in P–V diagram (not just endpoints). That’s why W is not a state function, unlike internal energy (U).
Isothermal vs Adiabatic
- Isothermal: Quasi-static process with thermal contact. ΔU = 0 (ideal gas). So, ΔQ = ΔW.
- Adiabatic: Insulated system (no heat exchange). ΔQ = 0 ⇒ ΔW = −ΔU. Temperature changes during expansion/compression.
Show Answer
W = nRT ln(V₂/V₁) = 1 × 8.31 × 300 × ln(2) ≈ 8.31 × 300 × 0.693 ≈ 1728 J.6. Reversible & Irreversible Processes
A process is reversible if both the system and surroundings can be restored to their original states with no other change in the universe.
Conditions for Reversibility
- Must be quasi-static (infinitely slow, always in equilibrium)
- Must have no dissipative effects (friction, viscosity, resistance)
Irreversible Processes (Most Natural Processes)
Irreversibility arises due to:
- Non-equilibrium states: Free expansion, explosive reactions
- Dissipative effects: Friction converts work to heat irreversibly
Examples: diffusion of gas, stirring liquid, cooling of hot object.
The Carnot engine is the only practical example of a reversible engine. All real engines are irreversible and less efficient.
7. Second Law & Heat Engines
The First Law allows energy conservation but doesn’t restrict the direction of processes. The Second Law introduces irreversibility and defines the natural direction of heat flow.
Statements of the Second Law
- Kelvin-Planck Statement: “No process is possible whose sole result is the absorption of heat from a reservoir and the complete conversion of the heat into work.” → Efficiency η < 100%.
- Clausius Statement: “No process is possible whose sole result is the transfer of heat from a colder object to a hotter object.” → Heat cannot spontaneously flow from cold to hot.
Heat Engine
A device that converts heat from a hot reservoir into useful work, rejecting some waste heat to a cold reservoir.
Where:
- Q₁ = heat absorbed from source at temperature T₁ (in Kelvin)
- Q₂ = heat rejected to sink at temperature T₂
8. Refrigerators & Heat Pumps
A refrigerator uses external work to extract heat from a cold interior and dump it into a warmer room.
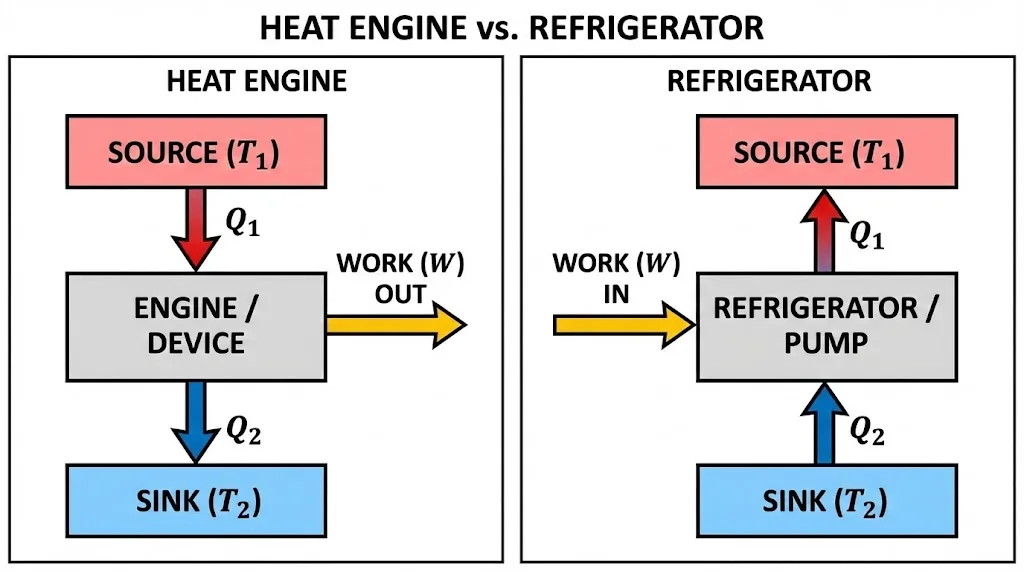
Coefficient of Performance (COP or α)
For a heat pump (used for heating), ![]() .
.
Show Answer
T₂ = 273 K (cold), T₁ = 300 K (hot).α = T₂ / (T₁ − T₂) = 273 / (300 − 273) = 273 / 27 ≈ 10.1.
9. Carnot Engine & Cycle
The Carnot engine is an idealized reversible heat engine operating between two temperatures. It gives the maximum possible efficiency for any heat engine working between those temperatures.
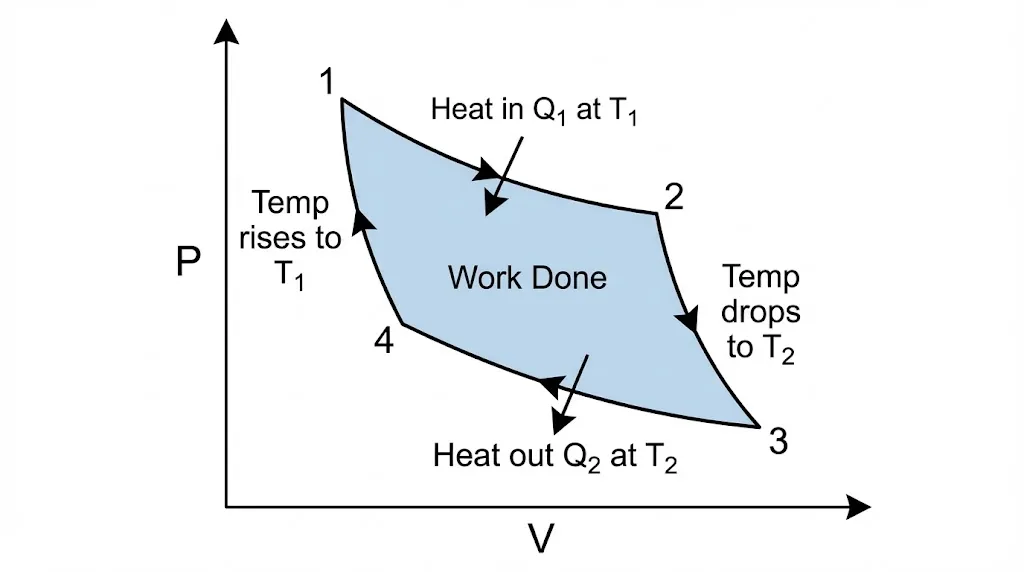
Four Reversible Stages
- Isothermal Expansion (A→B): Absorbs heat Q₁ at high temperature T₁.
- Adiabatic Expansion (B→C): Expands further; temperature drops from T₁ to T₂.
- Isothermal Compression (C→D): Rejects heat Q₂ at low temperature T₂.
- Adiabatic Compression (D→A): Compressed back; temperature rises from T₂ to T₁.
Derivation: Carnot Engine Efficiency η = 1 − T₂/T₁ 5 Marks • JEE Advanced
🔍 Show Full Derivation
Gas absorbs heat Q₁ from hot reservoir at temperature T₁.
Since isothermal:
No heat exchange (
For adiabatic process:
→
Gas rejects heat
(Note:
Temperature rises from
→
→
→


Since
→
→
• Efficiency depends only on temperatures, not on working substance
• T₁ and T₂ must be in Kelvin
• Maximum possible efficiency for any heat engine between T₁ and T₂
Carnot’s Theorem
- (a) No real engine operating between T₁ and T₂ can be more efficient than a Carnot engine.
- (b) The Carnot efficiency is independent of the working substance.
Show Answer
T₁ = 127 + 273 = 400 K. Q₁ = 1000 J, Q₂ = 600 J.Efficiency η = 1 − Q₂/Q₁ = 1 − 600/1000 = 0.4 = 40%.
Also, η = 1 − T₂/T₁ ⇒ 0.4 = 1 − T₂/400 ⇒ T₂ = 240 K = −33°C.
