Units and Measurement
NCERT Chapter 1 • SI Units, Significant Figures & Dimensional Analysis

1. Fundamental & Derived Units
Measurement involves comparing a physical quantity with an internationally accepted reference standard called a unit.
- Fundamental (Base) Units: The units for the base quantities which are independent of each other (e.g., Length, Mass, Time).
- Derived Units: Units expressed as combinations of base units (e.g., Velocity = Length/Time).
A complete set of these units is known as the System of Units.
(a) Kilogram (b) Second (c) Newton (d) Candela
Show Answer
Answer: (c) Newton. It is derived from kg·m·s⁻².Show Answer
Because they are not universally reproducible or based on invariant constants. SI units are defined via fundamental constants (e.g., speed of light), ensuring global consistency.Show Answer
1 Pa = 1 N/m² = (10⁵ dyne) / (10² cm)²= 10⁵ / 10⁴ dyne/cm²
= 10 dyne/cm².
2. The International System (SI)
The standard scheme used globally is the Système Internationale d’ Unites (SI). It consists of 7 Base Units and 2 Supplementary Units.
| Base Quantity | Unit Name | Symbol |
|---|---|---|
| Length | Metre | m |
| Mass | Kilogram | kg |
| Time | Second | s |
| Electric Current | Ampere | A |
| Temperature | Kelvin | K |
| Amount of Substance | Mole | mol |
| Luminous Intensity | Candela | cd |
Base units are now defined by fixing exact values of universal constants—e.g., the metre is defined using the speed of light (c = 299,792,458 m/s), and the kilogram via the Planck constant (h).
Supplementary Units (Plane & Solid Angle)
Besides the base units, SI defines two dimensionless units:
- Plane Angle (
 ): Ratio of arc length to radius. Unit: Radian (rad).
): Ratio of arc length to radius. Unit: Radian (rad). - Solid Angle (
 ): Ratio of intercepted area to square of radius. Unit: Steradian (sr).
): Ratio of intercepted area to square of radius. Unit: Steradian (sr).
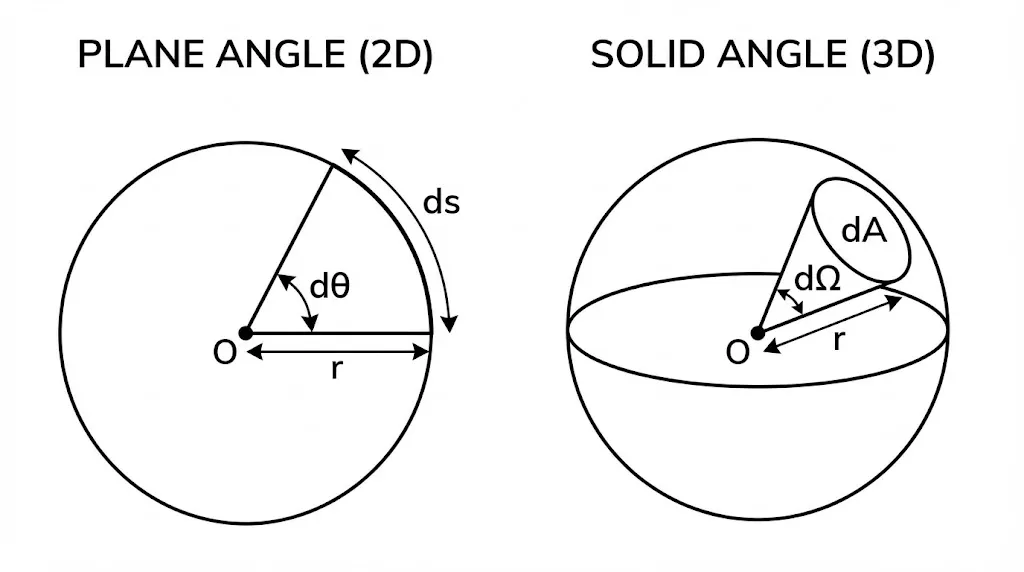
 ) vs. Solid Angle (
) vs. Solid Angle ( ).
).Show Answer
Yes. Both are ratios of same-dimensional quantities (length/length and area/area²), so their dimensions cancel out.3. Significant Figures
The reported result of a measurement is a number that includes all reliable digits plus the first uncertain digit. These are called Significant Figures.
A change of units does NOT change the number of significant figures.
Example: ![]() cm and
cm and ![]() m both have 4 significant figures.
m both have 4 significant figures.
Rules for Counting Significant Figures
- All non-zero digits are significant.
- Zeros between two non-zero digits are significant (e.g., 2005 has 4).
- Leading zeros (left of the first non-zero) are NOT significant (e.g., 0.007 has 1).
- Trailing zeros without a decimal are NOT significant (e.g., 12300 has 3).
- Trailing zeros WITH a decimal are significant (e.g., 3.500 has 4).
Scientific Notation: The Unambiguous Way
To eliminate confusion about trailing zeros, always use scientific notation:
4.700 m = 4.700 × 10³ mm → clearly has 4 significant figures.The power of 10 does not affect significant digit count.
The exponent in a × 10b gives the order of magnitude (e.g., Earth’s diameter ≈ 10⁷ m → order = 7).
Show Answer
(a) 3 (Leading zeros ignored, trailing zero counts), (b) 2 (Trailing zeros no decimal), (c) 4 (Scientific notation base counts).Show Answer
6.032 × 10⁻⁴ → 4 significant figures.4. Errors in Measurement
No measurement is perfect. The difference between the measured value and the true value is the error. We must also account for error propagation in calculations.
A. Arithmetic Operations with Significant Figures
Multiplication & Division: The final result should retain as many significant figures as the number with the least significant figures.
Mass = 4.237 g (4 sig figs)
Volume = 2.51 cm³ (3 sig figs)
Density =
Addition & Subtraction: The final result should retain as many decimal places as the number with the least decimal places.
Round to 663.8 g (1 decimal place).
B. Rounding Rules
• Round to the nearest even number for the preceding digit.
Examples:
– 2.745 → 2.74 (4 is even)
– 2.735 → 2.74 (3 is odd → round up)
1. Addition/Subtraction: Absolute errors add up. (![]() )
)
2. Multiplication/Division: Relative (%) errors add up. (![]() ).
).
3. Powers (![]() ): Relative error is multiplied by
): Relative error is multiplied by ![]() . (
. (![]() ).
).
Show Answer
AreaRelative Error
Absolute Error
Rounding to significant figures (since errors are 1 sig fig approx):
Area = (19.4 ± 1.5) cm².
Show Answer
% Error in
Show Answer
(a) 2.34 (Preceding digit 4 is even, drop 5).(b) 2.34 (Preceding digit 3 is odd, round up).
5. Dimensional Analysis
The dimensions of a physical quantity are the powers to which the base quantities are raised to represent that quantity.
We use square brackets: Length ![]() , Mass
, Mass ![]() , Time
, Time ![]() .
.
![Flowchart connecting physical quantities to fundamental dimensions Mass [M], Length [L], and Time [T].](/wp-content/uploads/2026/01/dimensional-analysis-flowchart.webp)
Velocity:
Force:
Energy:
Note: Pure numbers, ratios of same quantities (e.g., angle = arc/radius), and mathematical functions (sin, log, exp) are dimensionless.
Principle of Homogeneity
Physical quantities can only be added or subtracted if they have the same dimensions. This allows us to check the consistency of equations.
Let time period
Write dimensions for both sides:
Show Answer
All terms have dimension [LT⁻¹]. Equation is dimensionally consistent.Show Answer
Using dimensional analysis:Show Answer
LetEquating powers:
Formula:
