Kinetic Theory
NCERT Chapter 12 • Ideal Gas, Pressure, Equipartition of Energy & Mean Free Path

1. Introduction & Historical Context
In 1661, Robert Boyle discovered the law named after him. Boyle, Newton and several others tried to explain the behaviour of gases by considering that gases are made up of tiny atomic particles. The actual atomic theory got established more than 150 years later. Kinetic theory explains the behaviour of gases based on the idea that the gas consists of rapidly moving atoms or molecules. This is possible as the inter-atomic forces, which are short range forces that are important for solids and liquids, can be neglected for gases. The kinetic theory was developed in the nineteenth century by Maxwell, Boltzmann and others. It has been remarkably successful. It gives a molecular interpretation of pressure and temperature of a gas, and is consistent with gas laws and Avogadro’s hypothesis. It correctly explains specific heat capacities of many gases. It also relates measurable properties of gases such as viscosity, conduction and diffusion with molecular parameters, yielding estimates of molecular sizes and masses.
2. Molecular Nature of Matter
Richard Feynman, one of the great physicists of 20th century considers the discovery that “Matter is made up of atoms” to be a very significant one…
Atomic Hypothesis: All things are made of atoms—little particles that move around in perpetual motion, attracting each other when they are a little distance apart, but repelling upon being squeezed into one another.
Ancient Ideas: Kanada (India, 6th century BCE) proposed Paramanu. Democritus (Greece, 4th century BCE) coined “atom” (indivisible).
3. Behaviour of Gases & Ideal Gas Equation
Gases at low pressures and high temperatures much above that at which they liquefy approximately satisfy:
Where R = 8.314 J/mol·K, kB = 1.38 × 10⁻²³ J/K, NA = 6.02 × 10²³ mol⁻¹.
At STP, 1 mole occupies 22.4 litres.
Dalton’s Law of Partial Pressures: P = P₁ + P₂ + …
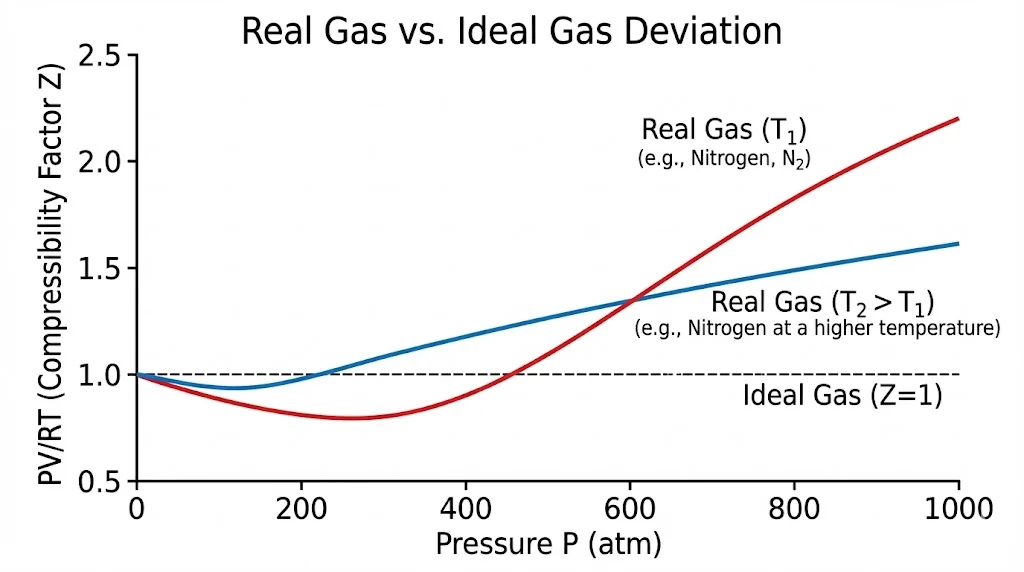
3A. Real Gases vs. Ideal Gases
Real gases obey the Ideal Gas Law only at low pressure and high temperature. Deviations occur because:
- Intermolecular forces are not negligible at high pressure.
- Volume of molecules is not negligible compared to container volume.
Van der Waals Equation (for real gases):
where a accounts for intermolecular attraction and b for excluded volume.
4. Pressure of an Ideal Gas
Pressure arises from momentum transfer during molecular collisions with walls.
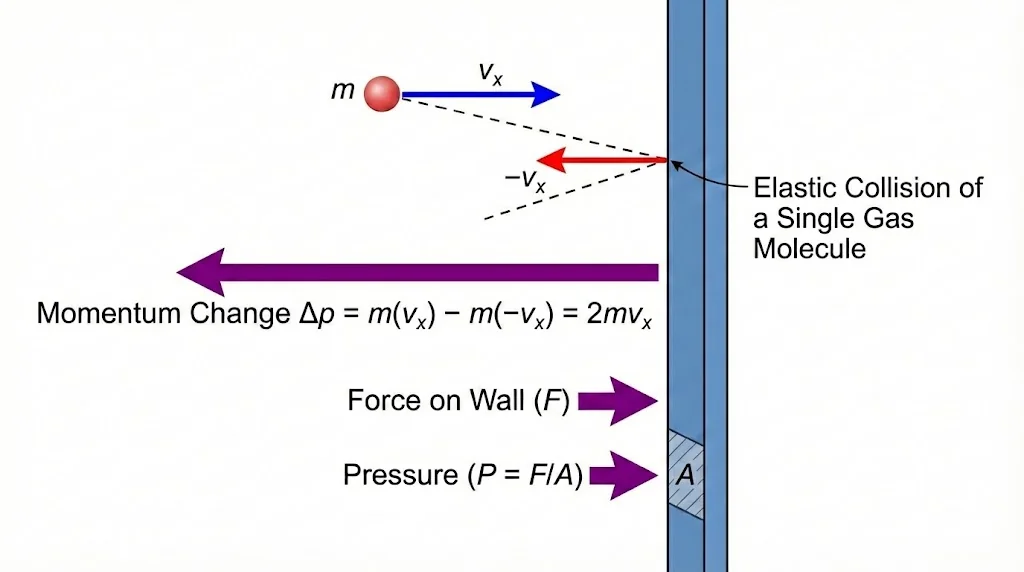
A molecule (mass m, x-velocity vx) hits wall elastically.
Δp = –mvx – (mvx) = –2mvx.
Momentum imparted to wall = +2mvx.
In time Δt, molecules within distance vxΔt hit wall.
Volume = A vx Δt → Number hitting = ½ n A vx Δt.
Total momentum transferred = (½ n A vx Δt)(2m vx) = n m A vx² Δt.
Force F = Δp/Δt = n m A vx².
P = F/A = n m vx².
Due to randomness: ⟨vx²⟩ = ⟨vy²⟩ = ⟨vz²⟩ = ⅓ ⟨v²⟩.
P = ⅓ n m ⟨v²⟩ = ⅓ ρ ⟨v²⟩
4A. Deduction of Gas Laws from Kinetic Theory
We know from kinetic theory: PV = ⅔ E, where E is total translational kinetic energy.
- Boyle’s Law: If T is constant, average KE is constant ⇒ E constant ⇒ PV = constant.
- Charles’ Law: Since E ∝ T, at constant P: V ∝ T.
- Avogadro’s Law: For two gases at same P, V, T:
⅓ (N₁/V) m₁⟨v₁²⟩ = ⅓ (N₂/V) m₂⟨v₂²⟩.
But ½ m₁⟨v₁²⟩ = ½ m₂⟨v₂²⟩ (same T) ⇒ N₁ = N₂.
5. Kinetic Interpretation of Temperature
From PV = ⅓ N m ⟨v²⟩ and PV = N kB T:
Conclusion: The average translational kinetic energy of a molecule is proportional to absolute temperature and independent of the gas type.
Root Mean Square (RMS) Speed
Show Answer
T = 300 K, M = 32 × 10⁻³ kg/mol.vrms = √(3 × 8.314 × 300 / 0.032) = √(233831) ≈ 484 m/s.
5A. Maxwell-Boltzmann Speed Distribution
Molecules in a gas do not all move with the same speed. Maxwell and Boltzmann derived a distribution curve showing how many molecules have a specific speed at a given temperature.
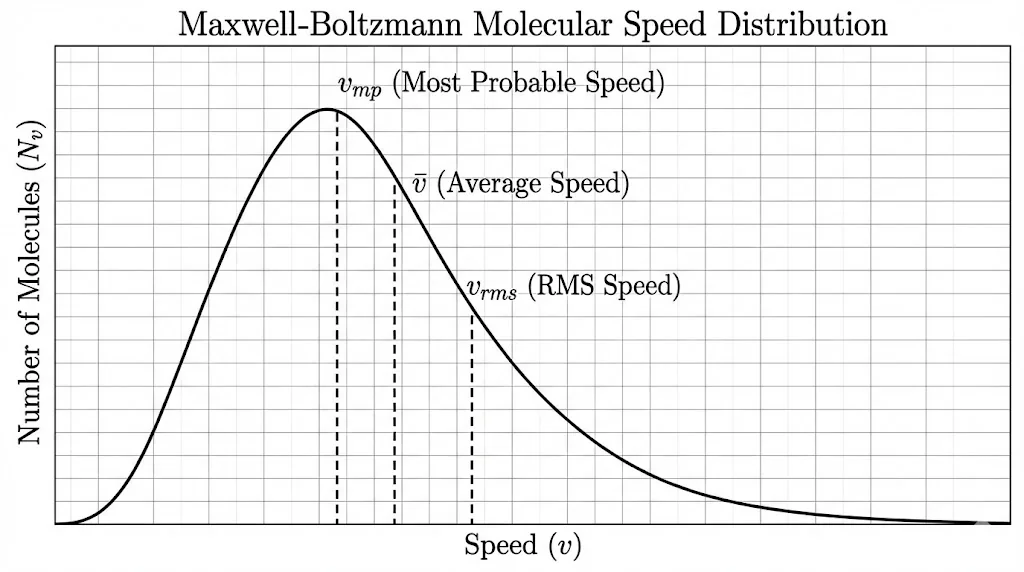
Different Types of Speeds
- Most Probable Speed (vmp): The speed possessed by the maximum number of molecules.
vmp = √(2RT/M) - Average Speed (v̄): Arithmetic mean of speeds.
v̄ = √(8RT/πM) ≈ 1.59 √(RT/M) - RMS Speed (vrms): Root mean square speed.
vrms = √(3RT/M) ≈ 1.73 √(RT/M)
6. Law of Equipartition of Energy
In thermal equilibrium, energy is equally distributed among all quadratic degrees of freedom. Each degree contributes ½ kBT per molecule.
| Type | f (Total) | U (per mole) | Cv | γ |
|---|---|---|---|---|
| Monatomic | 3 | 3/2 RT | 3/2 R | 5/3 ≈ 1.67 |
| Diatomic (rigid) | 5 | 5/2 RT | 5/2 R | 7/5 = 1.40 |
| Diatomic (vibrating) | 7 | 7/2 RT | 7/2 R | 9/7 ≈ 1.29 |
| Polyatomic (non-linear) | 6+ | 3RT+ | 3R+ | <1.33 |
7. Specific Heat Capacity
7.1 Gases
From equipartition: Cv = f/2 R, Cp = Cv + R, γ = 1 + 2/f.
7.2 Mixture of Non-Reacting Gases
For a mixture of ![]() moles of Gas 1 and
moles of Gas 1 and ![]() moles of Gas 2:
moles of Gas 2:
The specific heat ratio for the mixture is ![]() .
.
Show Answer
He:7.3 Solids (Dulong-Petit Law)
Each atom vibrates in 3D → 6 degrees → U = 3RT per mole.
Show Answer
Volume = 44.8 L = 2 moles.He is monatomic → Cv = 3/2 R.
Q = n Cv ΔT = 2 × (3/2 × 8.314) × 15 ≈ 374 J.
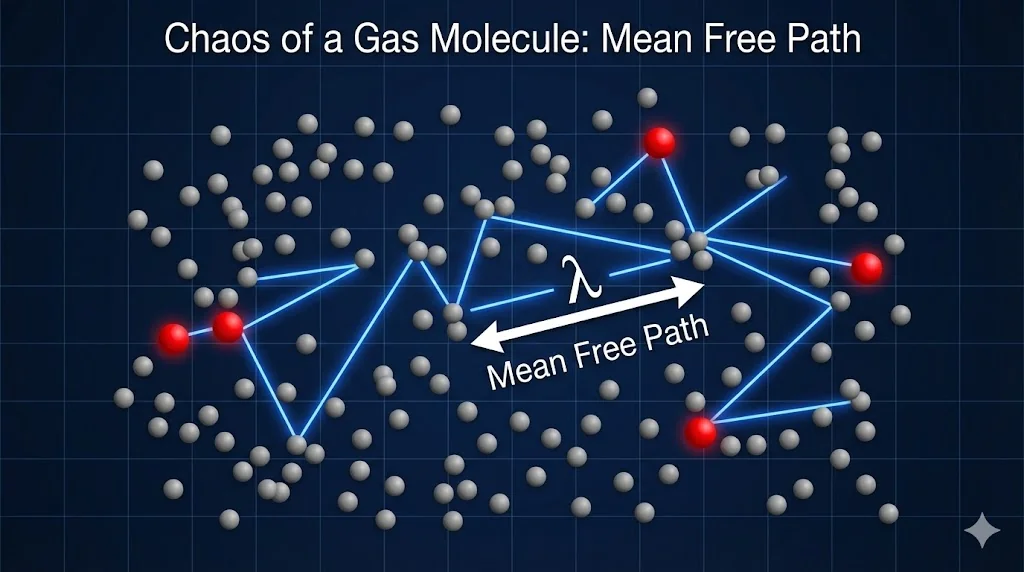
8. Mean Free Path (λ)
The average distance a molecule travels between two successive collisions.
In uranium enrichment, lighter 235UF6 molecules have higher vrms and effuse faster through porous barriers.
