Advanced Physics → Advanced Thermal Physics → Real Gases & Deviations
• Ideal gas assumptions break down at high pressure and low temperature
• Real gases exhibit intermolecular attraction and finite molecular volume
• van der Waals equation corrects the ideal gas law
1. Why Ideal Gas Theory Fails
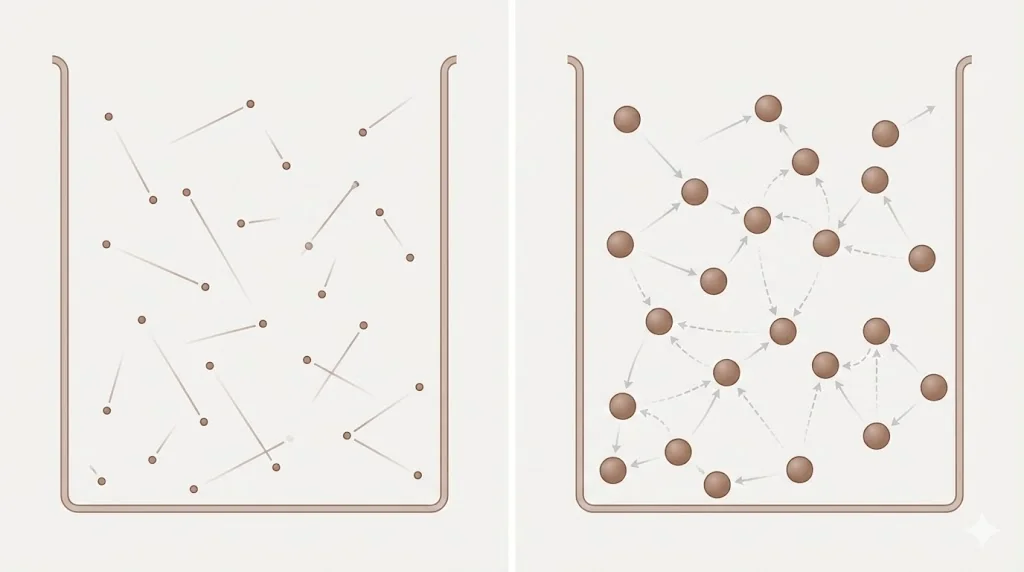
The kinetic theory of gases treats molecules as point particles with no intermolecular forces except during elastic collisions.
This approximation works well only at low pressure and high temperature.
These deviations arise from two fundamental microscopic effects.
2. Two Fundamental Causes of Deviation
(a) Intermolecular Forces
At moderate separations, gas molecules attract each other due to van der Waals forces.
This attraction reduces the effective pressure exerted on the container walls.

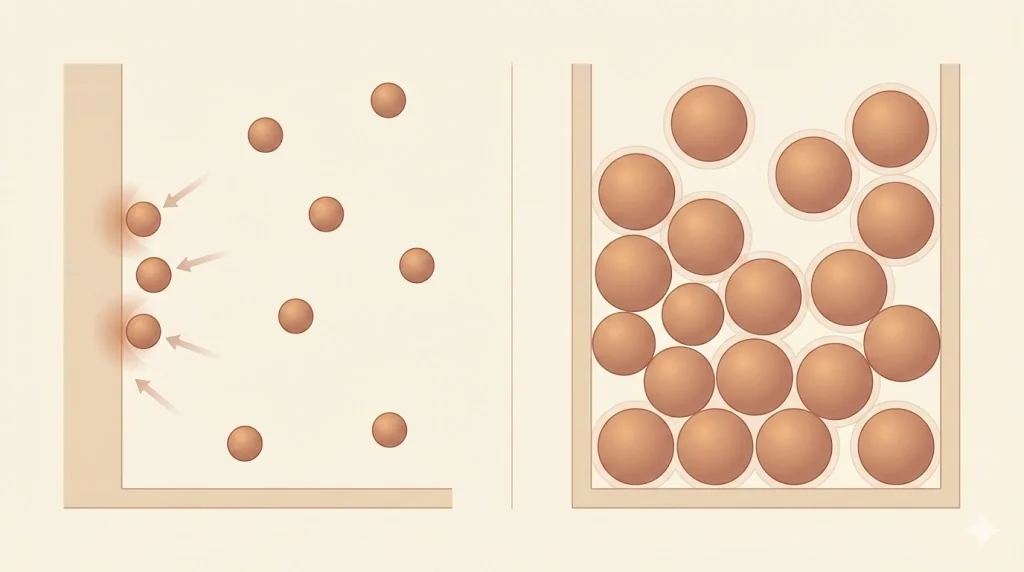
(b) Finite Molecular Size
Molecules occupy finite volume. At high pressure, the free volume available for motion is significantly reduced.

3. van der Waals Corrections
Pressure Correction
![]()
Volume Correction
![]()
4. van der Waals Equation of State
![]()
- a measures intermolecular attraction
- b accounts for finite molecular volume
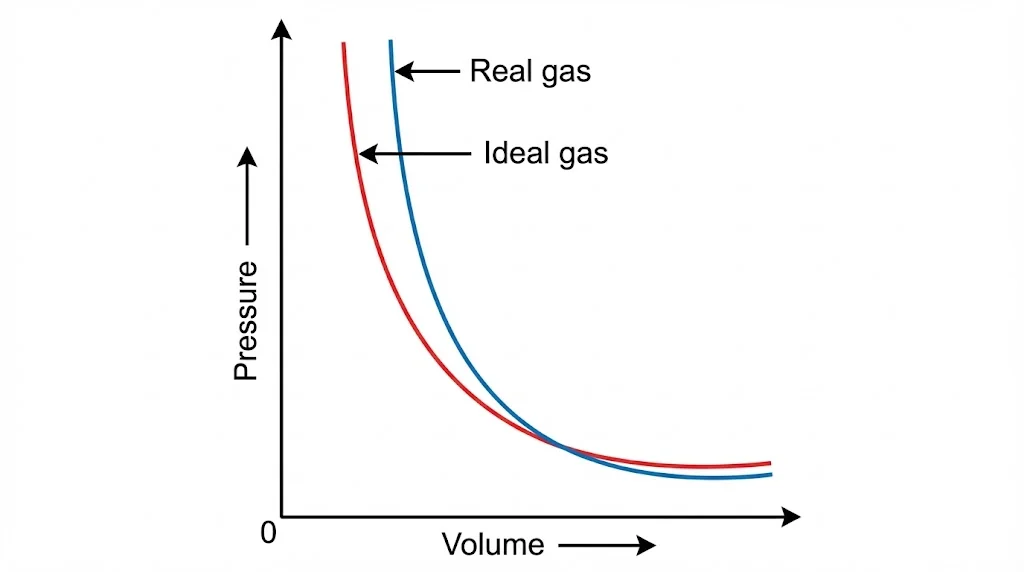
5. Physical Interpretation
At low density, correction terms become negligible and the ideal gas law is recovered.
At high density, deviations become significant and ideal gas theory fails completely.

6. Critical Phenomena
When a real gas is compressed and cooled, the distinction between gas and liquid gradually disappears. The specific temperature and pressure at which this happens are called the critical temperature and critical pressure.
At the critical point, the system undergoes a profound change: large-scale density fluctuations appear, and the concept of a sharp gas–liquid boundary loses meaning.
Near this point, the gas becomes highly compressible. Small changes in pressure or temperature produce unusually large changes in density. This behavior cannot be explained using the ideal gas model.
From the perspective of van der Waals theory, the critical point corresponds to an inflection point on the pressure–volume isotherm:
![]()
Physically, this means that the restoring tendency of pressure against volume change vanishes. As a result, the system becomes unstable against density fluctuations.
Critical phenomena reveal a deep limitation of classical thermodynamics. Near the critical point, mean-field models like the van der Waals equation fail to predict experimental behavior accurately. A full understanding requires ideas from statistical mechanics and scaling theory.
7. Where Real Gas Theory Appears Next
- Liquefaction of gases
- Critical phenomena
- Statistical mechanics
- Phase transitions
